
Making smoking healthier and more personalized, the JR10 e-cigarette is specially designed for the UK market and future European markets. With its excellent design and attractive appearance, it stands out as a leader among smoking alternatives.

Core Highlights: Long-lasting Battery Life and Powerful Performance
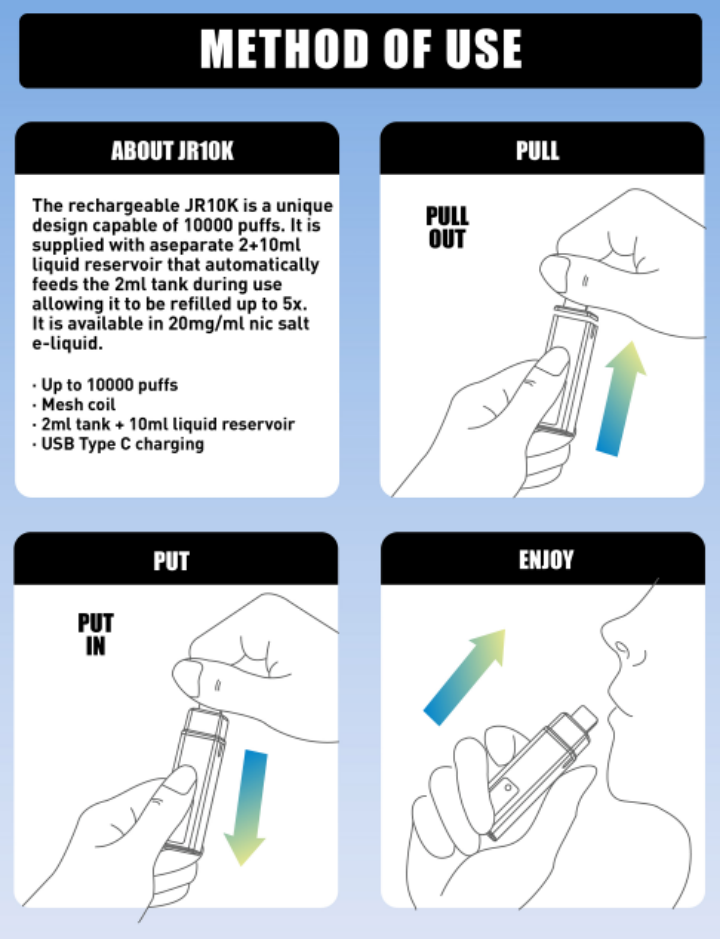
The JR10 is equipped with a 650mAh rechargeable battery, supporting USB-C fast charging. It can be fully charged in less than an hour, allowing you to maintain smooth use whether at home, in the office, or on the go. Coupled with a large 2+10ML semi-transparent cartridge, it supports approximately 10,000 draws, truly achieving a perfect balance of efficiency and long-lasting battery life.
Imagine: No need for frequent charging and oil refills, easily meeting the needs of the whole day, making your life more convenient and efficient.

Colorful Lights Add Fun to the Experience
Every draw lights up the semi-transparent cartridge with a rainbow of colors, providing a cool visual experience. This is not just a function but also a fashion statement, suitable for modern users who pursue personalization.
Automatic Airflow Activation Mechanism, Mouth-to-Lung (MTL) Style for a More Natural Fit
The JR10 adopts an automatic airflow activation mechanism, no button operation is needed, just pick up and use, especially suitable for users who prefer the MTL (mouth-to-lung) drawing style, with a smooth and natural draw.
Details are King: Thoughtful Design for Every Moment
- Battery Level Screen Display: Always in control of the device status, no need to worry about the battery running out suddenly.
- 20mg Nicotine Concentration: Precise control of nicotine cravings, meeting needs while complying with the strict regulations of the European market.
- Semi-transparent cartridge Design: Clear oil level visibility, more worry-free use.
Born for the Future: A Dual Commitment to Safety and Environmental Sustainability
As an innovative product focused on the UK and European markets, the JR10 not only complies with local laws and health standards but also offers sustainable choices through more environmentally friendly designs.
The above 2+10 product is developed by Lanavape, which has not yet been officially launched on the website.
LANAVAPE is a well-known e-cigarette brand factory headquartered in Shenzhen, China, ranking in the top five in the Southeast Asian market and trusted by consumers. The company focuses on the research and production of disposable, Replaceable pod, and open pod-style e-cigarettes. Now, LANAVAPE is actively expanding into the European market, offering healthier and more efficient smoking alternatives to users worldwide.










