In June 2022, the Philippines Department of Trade and Industry (DTI) issued Department Administrative Order (DAO) No. 22-06, introducing new technical regulations mandating product certification for vaporized nicotine and non-nicotine products, including electronic cigarettes, in order to enforce compliance with manufacturing, distribution, and sales standards in the Philippine market.
Local and foreign manufacturers selling/distributing e-cigarettes in the Philippines market should participate in the PS Safety and Quality Certification Scheme (also known as the PS Licensing Scheme). The governing body grants qualifying companies a license to use the PS certification mark.
To ensure the conformity of certified products, regulatory agencies should conduct regular monitoring activities in accordance with current DTI rules. This includes annual factory system and product audits, as well as random product audits in local companies holding valid PS licenses, and regular product audits for each shipment in foreign companies holding valid PS licenses.
Controlled product range includes: a. Completed electronic cigarette products (vapor products); b. Electronic cigarette devices used for vaporizing products; c. Completed heated tobacco products (HTP).
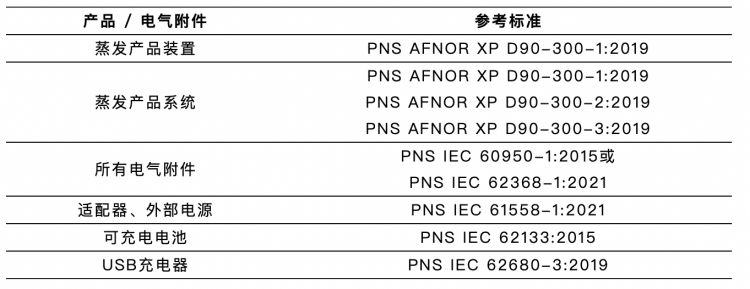
Product conformity requirements: a. Products must be tested by the testing laboratory of the Philippine Bureau of Standards (BPS) or other BPS-recognized testing laboratories to validate their conformity. b. Applicable products and related electrical accessories must meet the standard requirements of the reference standards.
C. The evaporative product facility/system must also comply with the migration testing requirements of Regulation (EU) 10/2011 and (EU) 20/1245; D. The embossed/engraved/printed identification must be tested to ensure clarity and durability; E. This also includes a factory inspection.
The PS certification mark should comply with Design A for steam product systems and heating tobacco product systems that have been certified to meet safety and performance requirements. For steam product devices certified to meet safety requirements, the PS mark should comply with Design B. The size of the PS mark attached to the product or its packaging may be enlarged or reduced as long as it complies with the specifications in Design A or B.
Effective/Implementation Date.
a. The DAO will officially come into effect 15 days after its announcement. b. The regulating authority must enforce and monitor the DAO within 30 months of its implementation date. After this date, only vaporized nicotine and non-nicotine products with valid PS, ICC or other BPS certification marks can be sold in the local market.
The Philippines' issuance of Administrative Order No. 22-06 signifies another step towards regulatory oversight of new tobacco products. Electronic cigarette manufacturers for export must apply for Product Standards (PS) and Import Commodity Clearance (ICC) within 18 months. Products already being sold in the local market are advised to apply for certification within 12 months of the DAO's implementation.
Statement:
This article is compiled from third-party information and is intended for industry communication and learning purposes only.
This article does not represent the views of 2FIRSTS, and 2FIRSTS is unable to confirm the authenticity and accuracy of its contents. The compilation of this article is intended solely for industry discussions and research.
Due to limitations in translation abilities, the translated article may not fully represent the content of the original. Please refer to the original text for accuracy.
2FIRSTS maintains complete alignment with the Chinese government on all statements and positions involving domestic affairs, Hong Kong, Macau, Taiwan, and foreign affairs.
Copyright of the compiled information belongs to the original media and author. If there is any infringement, please contact us to have it removed.
This document has been generated through artificial intelligence translation and is provided solely for the purposes of industry discourse and learning. Please note that the intellectual property rights of the content belong to the original media source or author. Owing to certain limitations in the translation process, there may be discrepancies between the translated text and the original content. We recommend referring to the original source for complete accuracy. In case of any inaccuracies, we invite you to reach out to us with corrections. If you believe any content has infringed upon your rights, please contact us immediately for its removal.









