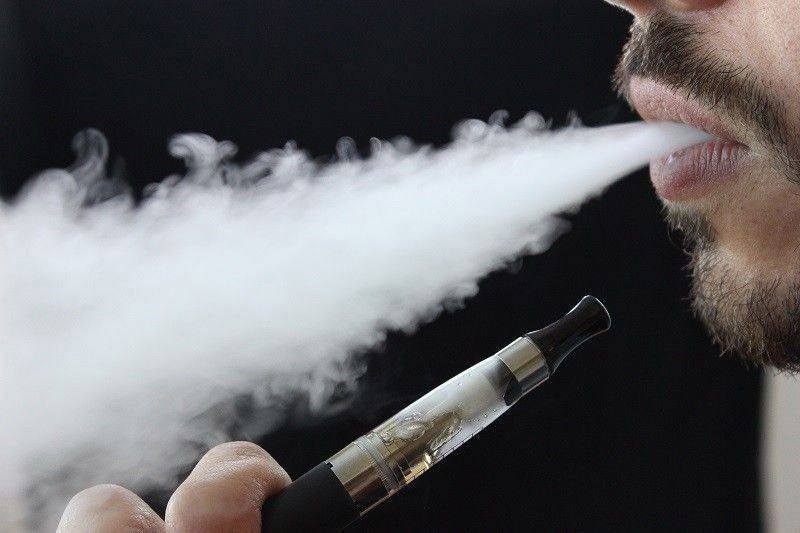
This week, Multnomah County once again made efforts to ban flavored nicotine products, including flavored e-liquids and menthol cigarettes. The ban was first considered by the county commissioners in 2019, but was delayed due to the pandemic.
The possible ban aims to minimize the onset of nicotine addiction among adolescents. According to a 2015 study - the latest data available - the majority of Americans aged 12 to 17 have experimented with tobacco or nicotine, starting with flavored products. 81% of them reported using flavored e-cigarettes while 50% reported using flavored cigarettes, such as menthol or clove.
According to data from the Oregon Health Authority, the use of nicotine products among young people is becoming increasingly common. In 2013, one in ten high school students in Oregon reported using e-cigarettes. By 2015, this number had risen to one in six. Nationally, it is estimated that approximately one in four high school students in the US currently use e-cigarettes - devices that release nicotine in vapor form, rather than burning tobacco like traditional cigarettes. The data also shows that the vast majority of these young smokers use flavored nicotine products, with 80% of 12-17 year old smokers reporting use of flavored products, and 29% of smokers over the age of 65 reporting the same.
During her presentation to the board, Vines showcased several nicotine and tobacco products that imitate candy flavors and packaging, such as a bear-shaped gummy candy vape liquid packaged like the actual candy. Flavored nicotine products are readily available in Monte Noma County, with 99.6% of retailers selling at least one flavored product, and 73% of those offering promotional prices for flavored products. Vines provided an example of three chocolate and vanilla-flavored cigarillos or mini-cigars sold for 99 cents, which is cheaper than most candy bars.
In 2009, when the US Food and Drug Administration banned flavored cigarettes, it made an exception for menthol cigarettes, which Vine believes to be a "gateway product." According to data from the anti-smoking organization Truth Initiative, menthol cigarettes are predominantly used by young smokers, with 53% of 12 to 17 year old smokers using the product and 32% of smokers aged 35 to 49 using it. Due to targeted marketing efforts by the tobacco industry, menthol cigarettes are also heavily used by smokers of color, particularly African Americans in the US.
After a series of serious lung infections related to electronic cigarettes have occurred in Oregon and across the country, Multnomah County is considering banning flavored e-cigarette liquids and tobacco products by the end of 2019. Nationally, nearly 70 people have died from lung fungal infections related to electronic cigarettes. Despite the health risks associated with e-cigarettes, they are widely used as a means for smokers to quit using traditional cigarettes. As a doctor, Vines believes that transitioning heavy smokers to e-cigarettes is a step in the right direction, as they will no longer inhale toxic carcinogens present in cigarettes.
Without a doubt, combustible tobacco is worse for you than using e-cigarettes," Vines told the board. "This is especially true for adult heavy smokers who are trying to reduce harm by switching to e-cigarettes. However, the FDA has not yet approved e-cigarette products or e-liquids as smoking cessation aids." Vines noted that conclusive studies on the health effects of e-cigarettes are lacking due to the relatively new presence of nicotine-only products on the market. But Vines stated that the impact of nicotine use on the health of young people, whose brains are still developing, is concerning, especially when nicotine is combined with e-liquid. "My bottom line is that when a group of young people becomes addicted to nicotine, no one wins, whether they're using e-cigarettes or continuing to smoke combustible tobacco," Vines said.
Despite the disruption and delay caused by the COVID-19 pandemic, the county's plan to ban flavored nicotine and tobacco products continues to be a priority. The pandemic has highlighted the importance of addressing potential health conditions that make individuals more susceptible to the disease in the area. According to data from the American Heart Association, COVID patients who smoke have a 45% increased likelihood of death and a 39% increased likelihood of requiring a ventilator compared to non-smoking COVID patients. On Tuesday, the Multnomah County Chair, Deborah Kafoury, instructed county health officials to develop proposed policy language to prevent the sale of all flavored nicotine products before the end of September.
We know that many businesses in our community profit from these products, but sometimes we need to say enough," said Cafarelli. "After two years of deadly respiratory infections exacerbated by smoking, I am personally ready to say enough." If the ban moves forward, Motenoma County will become the second county in the state to ban flavored nicotine products. Washington County voters approved a ban on flavored tobacco, including menthol cigarettes, starting in 2022, but a judge temporarily overturned the ban in July, citing irreparable harm to convenience stores and hookah bars, among other businesses.
Opponents of the Washington County ban and the possible Multnomah County ban allege that the prohibition of flavored electronic cigarettes will lead smokers to revert back to traditional cigarettes, harm the sales of retail stores, and eliminate a choice in reducing the harm of smoking.
Statement
This article is compiled from third-party information and is intended solely for industry-related communication and learning purposes.
This article does not represent the views of 2FIRSTS and 2FIRSTS cannot confirm the authenticity and accuracy of the article's content. The translation of this article is only intended for industry research and communication purposes.
Due to limitations in our translation abilities, the translated article may not exactly reflect the original text. Please refer to the original article for accuracy.
2FIRSTS maintains complete alignment with the Chinese government on any domestic, Hong Kong, Macau, Taiwan, or foreign-related statements and positions.
The compilation of information is the property of the original media and author. If there is any infringement, please contact us for deletion.
This document has been generated through artificial intelligence translation and is provided solely for the purposes of industry discourse and learning. Please note that the intellectual property rights of the content belong to the original media source or author. Owing to certain limitations in the translation process, there may be discrepancies between the translated text and the original content. We recommend referring to the original source for complete accuracy. In case of any inaccuracies, we invite you to reach out to us with corrections. If you believe any content has infringed upon your rights, please contact us immediately for its removal.