Key Highlights:
● Acquirer: R.J. Reynolds Vapor Company (a subsidiary of British American Tobacco)
● Seller: Charlie’s Holdings, Inc.
● Transaction Date: April 16, 2025 (disclosed via SEC Form 8-K on April 17)
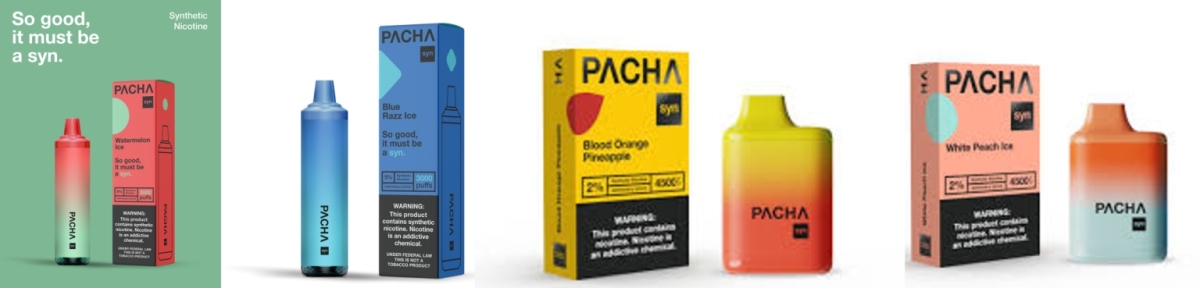
● Assets Sold: In 2022, 12 PACHA-brand synthetic nicotine products were submitted to the PMTA, all of which are disposable e-cigarettes
● Deal Value: $5 million upfront payment + up to $4.2 million in contingent earn-out
● Regulatory Status: Products submitted under FDA’s PMTA framework; approval status not disclosed
● Strategic Implication: BAT strengthens compliant product portfolio; Charlie’s monetizes regulatory-stage assets
● Market Response: CHUC stock surged 33.33% on April 17 following the announcement
[2Firsts] On April 17, 2025, Charlie’s Holdings, Inc. filed a Form 8-K with the U.S. Securities and Exchange Commission (SEC), announcing that it had entered into and completed an asset purchase agreement with R.J. Reynolds Vapor Company, a subsidiary of British American Tobacco (BAT), on April 16.
The deal involves the sale of 12 synthetic nicotine disposable e-cigarette products under the PACHA brand, all of which were submitted to the U.S. Food and Drug Administration (FDA) for PMTA review in 2022. The total consideration includes a $5 million upfront cash payment, plus a one-time contingent earn-out of up to $4.2 million, based on the volume of sales generated during the first 12 months of commercialization.
The agreement contains standard representations, warranties, and indemnities, and is filed as Exhibit 10.1 to the Form 8-K.
Following the announcement, Charlie’s Holdings (ticker: CHUC) surged 33.33% on April 17, closing at $0.10 per share—its highest closing price in nearly two months. The market response underscores strong investor confidence in the transaction and the monetization potential of Charlie’s regulatory-stage assets.
The 12 products sold are synthetic nicotine offerings currently under PMTA review, and it remains unclear whether any have received Marketing Granted Orders (MGOs) from the FDA. For BAT, the acquisition marks another step in expanding its portfolio of regulatory-stage products. For Charlie’s, it may signal a broader effort to streamline its operations or reallocate capital.
With intensifying regulation across the U.S. nicotine market, PMTA-submitted products are increasingly seen as valuable strategic assets. More acquisitions of this nature are likely to follow.