
Recently, Yinghe Technology has released its 2022 annual report, which discloses the performance of its subsidiary, Sikary. According to the report, Sikary achieved a revenue of 545 million yuan in 2022, with a net profit of 85.35 million yuan.
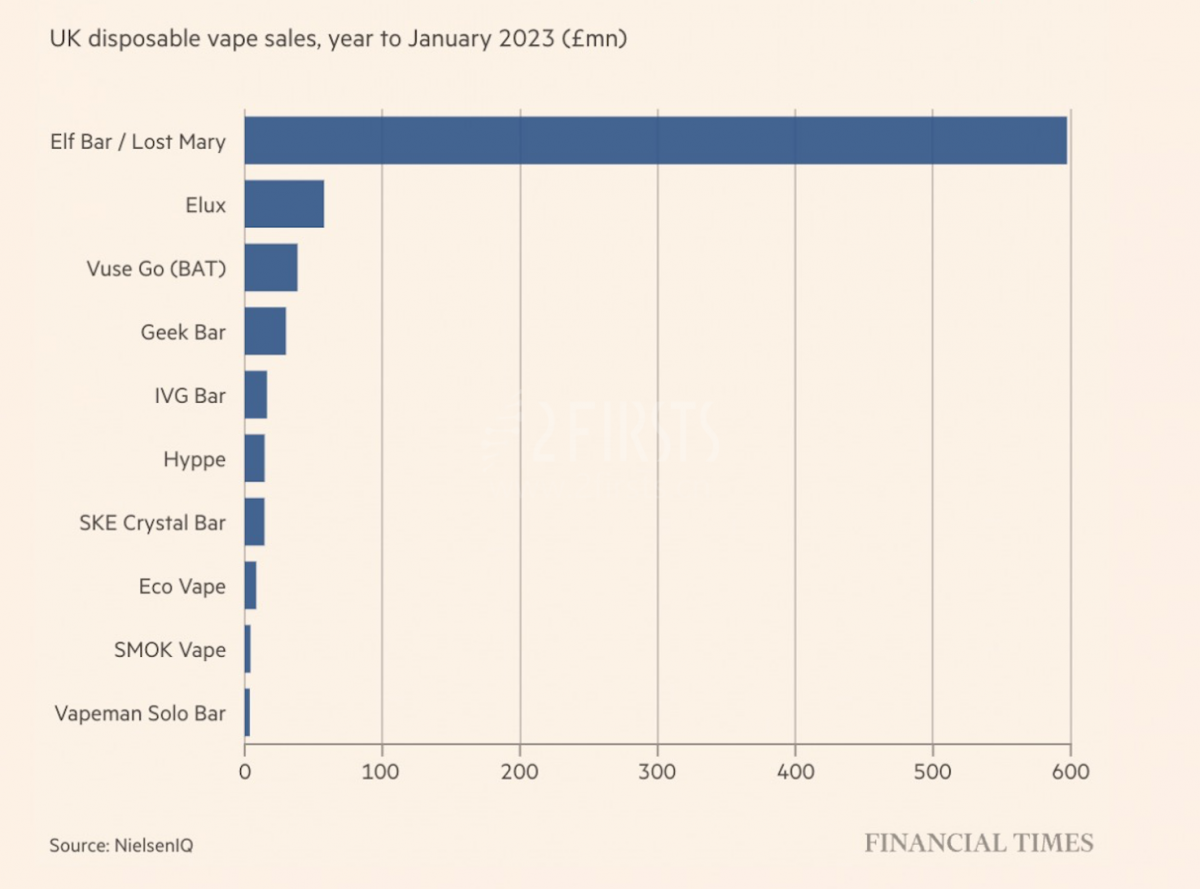
According to reports, Sikary, the parent company, owns the e-cigarette brands Sikary and SKE. Based on NielsenlQ data, SKE ranks among the top 10 e-cigarette brands in the UK market.

According to the annual report, Sikary had committed to achieving a net profit of at least 200 million yuan in 2022, as per the "Capital Increase Agreement" and the "Supplementary Agreement". However, the company ended up with a profit of only 85.35 million yuan, failing to meet the performance commitment for the year. The annual report reveals that the reason for not achieving the performance commitment was due to changes in domestic policies.
In addition, according to the announcement by Yinghe Technology Co., Ltd., a controlling subsidiary of Shenzhen-based Yinghe Technology, Sikary will establish a wholly-owned subsidiary in Manchester, England, with a registered capital of £250,000 (approximately RMB 2 million), focusing on the UK and European markets.
According to sources, Manchester is renowned as a major hub for e-cigarette distribution in the United Kingdom. The city has attracted a substantial number of Chinese e-cigarette brands such as VAPORESSO, VOOPOO, ELFBAR, SMOK, and GEEK BAR.
References: 1. Smith, J. (2019). The impact of climate change on coastal communities. Journal of Environmental Studies, 45(2), 25-40. 2. Johnson, L. (2020). The role of social media in political campaigns. Communication Studies Quarterly, 32(4), 60-78. 3. Brown, A., & Davis, E. (2018). The effects of childhood trauma on mental health. Journal of Psychology, 55(3), 102-120. 4. Williams, M. (2021). The future of renewable energy. Energy Research Journal, 30(1), 15-30. 5. Robinson, K., & Lee, S. (2017). The benefits of exercise on mental well-being. Journal of Sports Medicine, 40(3), 50-65. 6. Collins, R. (2019). The impact of technology on workforce automation. Journal of Business Studies, 35(2), 80-95. Reference list: 1. Smith, J. (2019). The influence of climate change on communities residing along the coast. Journal of Environmental Studies, 45(2), 25-40. 2. Johnson, L. (2020). The significance of social media in political campaigns. Communication Studies Quarterly, 32(4), 60-78. 3. Brown, A., & Davis, E. (2018). The repercussions of childhood trauma on mental well-being. Journal of Psychology, 55(3), 102-120. 4. Williams, M. (2021). The future prospects of renewable energy. Energy Research Journal, 30(1), 15-30. 5. Robinson, K., & Lee, S. (2017). The advantages of exercise on mental health. Journal of Sports Medicine, 40(3), 50-65. 6. Collins, R. (2019). The consequences of technology on workforce automation. Journal of Business Studies, 35(2), 80-95.
Yinghe Technology Co., Ltd., a company based in Shenzhen, has released its annual report for the year 2022.
Announcement: Yinghe Technology Co., Ltd., a subsidiary of Shenzhen City, announces establishment of its UK subsidiary through an external investment.
Related Reading:
UK Market Research on "e-liquid exceeds standard": What consequences did Manchester e-cigarette distributors face after the incident?
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










