
On September 19th, 2024, the InterTabac tobacco exhibition in Dortmund, Germany officially opened. On the opening day, 2Firsts conducted an exclusive interview with the popular e-cigarette brand FUMOT.
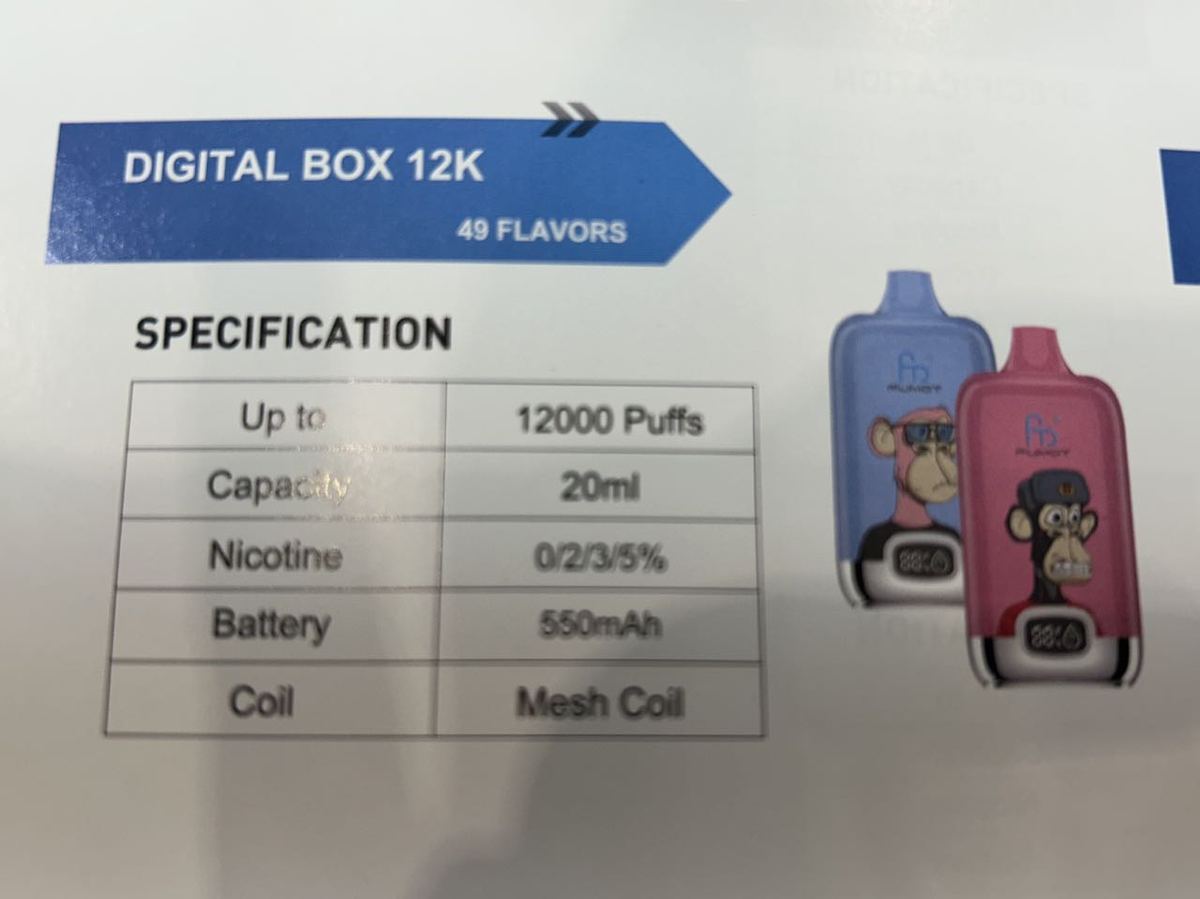
FUMOT staff briefed 2Firsts on the current situation in the German market. According to their information, leading brands in the compliant German market include ELFBAR, BLU, HQD, and FLERBAR. Additionally, FUMOT's flagship products have achieved shipments of several million units in the German market, making it their key market.

When discussing the mainstream product types in the German market, the staff member stated that due to factors such as low cost-effectiveness, small-sized disposable products have seen a significant decrease in market share in Germany, becoming increasingly marginalized. Conversely, new large-sized disposable products that comply with TPD standards are emerging, such as innovative products like "n in 1" and "2+10." Therefore, at this trade show, FUMOT also introduced a variety of innovative products like "2+10" to meet the demands of the German market and solidify its market position.
For brands looking to enter the German market, the staff member believes that although Germany is an important market in Europe with promising prospects, the acceptance of new brands is relatively low. Distributors tend to prefer familiar brands. Therefore, achieving success in the German market requires long-term cultivation and careful planning.
When it comes to regulation, the above-mentioned staff shared the regulatory situation of the e-cigarette market in Germany. According to their information, Germany imposes high taxes on e-liquid products, with a tax of 2 euros per compliant 10ml e-liquid. German police also conduct strict inspections of tax labels attached to products at the retail end. Among them, Berlin has the highest inspection frequency in the market, but the penalties for violations are relatively light, usually limited to confiscation of goods or fines. In contrast, the market in Cologne has the strictest regulation, and sellers of tax evasion products may face criminal liability.
According to reports, despite this, there are still underground markets in Germany, but these markets mainly operate through online channels such as WhatsApp and Instagram. Due to the high risks involved in offline transactions, only a few retail stores dare to get involved, and they usually only sell to trusted customers.
In the interview, FUMOT's onsite staff repeatedly mentioned the keyword "compliance." They believe that with the increasing strictness of market supervision in Germany, brands must actively align with compliance standards in order to achieve long-term stable development.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










