
Recently, the United States Senate officially confirmed Marty Makary as the 27th Commissioner of the Food and Drug Administration (FDA) with a vote of 56 in favor and 44 against. This nomination was put forth by then President Donald Trump on November 22, 2024, and ultimately approved by Congress, making Makary the top official at the FDA.
As the new leader of the FDA, Marty Makary will directly impact regulatory policies in the United States for food, drugs, medical devices, and new tobacco products. What is his background? What is his stance on new tobacco products, especially e-cigarettes? How will he lead the FDA? These are the questions currently receiving high attention from the industry and the public.

Renowned surgeon and bestselling author
Based on multiple sources, Makary is a renowned surgeon and bestselling author. He obtained his medical doctorate from Jefferson Medical College and a master's degree in public health from Harvard University School of Public Health early in his career. He later served as a surgical professor and director of pancreatic transplant surgery at Johns Hopkins University, as well as the director of the Minimally Invasive Pancreatic Surgery program at Johns Hopkins Hospital.
What really made him gain popularity among the public was his active involvement in researching medical policies, patient safety, and medical ethics, as well as his participation in developing the "Surgical Safety Checklist" aimed at reducing surgical errors. The checklist was promoted globally, enhancing public trust in his professional capabilities. In particular, his research and advocacy on medical policies, as evidenced by his bestselling books "The Price We Pay" and "Unaccountable," earned him widespread recognition. "The Price We Pay" even won the 2020 Business Book of the Year Award from the Business Journalists Association for its exploration of the injustices and systemic issues within the American healthcare system. In 2023, the book was introduced to China.

As a frontline surgeon, Makary gained first-hand knowledge of patient care through clinical practice, while his public health research and policy work provided him with a systematic understanding of healthcare challenges. Additionally, through his continuous writing, he has become a "celebrity" in the field. Trump's selection of him as the new head of the FDA highlights his desire to "restore the American public's trust in the FDA and address harmful chemicals in the nation's food supply and adolescent drugs and biological products.

The new position is approved by both houses in the end

On November 22, 2024, when Trump announced the nomination of Makary as FDA Commissioner, the public's impression of him remained focused on his identity as a surgeon. However, it is worth noting that Makary's direct superior is Health and Human Services (HHS) Secretary Robert F. Kennedy, Jr., who has garnered attention in the U.S. media for promoting anti-vaccine information and public health theories. Thus, the public's concerns about the U.S. healthcare system under his leadership have indirectly influenced their views on Makary.
On March 6, 2025, Makary attended a confirmation hearing held by the Senate Health, Education, Labor, and Pensions Committee (HELP). During the hearing, he repeatedly pledged to follow "scientific procedures" at the FDA. He also outlined his priorities, including a greater focus on chronic diseases such as diabetes and obesity. Additionally, he promised to review the staffing situation at the FDA, as well as the agency's operational capacity following recent layoffs. Regarding vaccines, Makary stated clearly that "vaccines can save lives," and mentioned the need for the agency to be more open to scientific dissent.
On March 25th, the Senate officially confirmed Makary's appointment to the FDA position with 56 votes in favor and 44 votes against. It is worth noting that three Democratic senators also voted in favor, making him the official FDA commissioner.
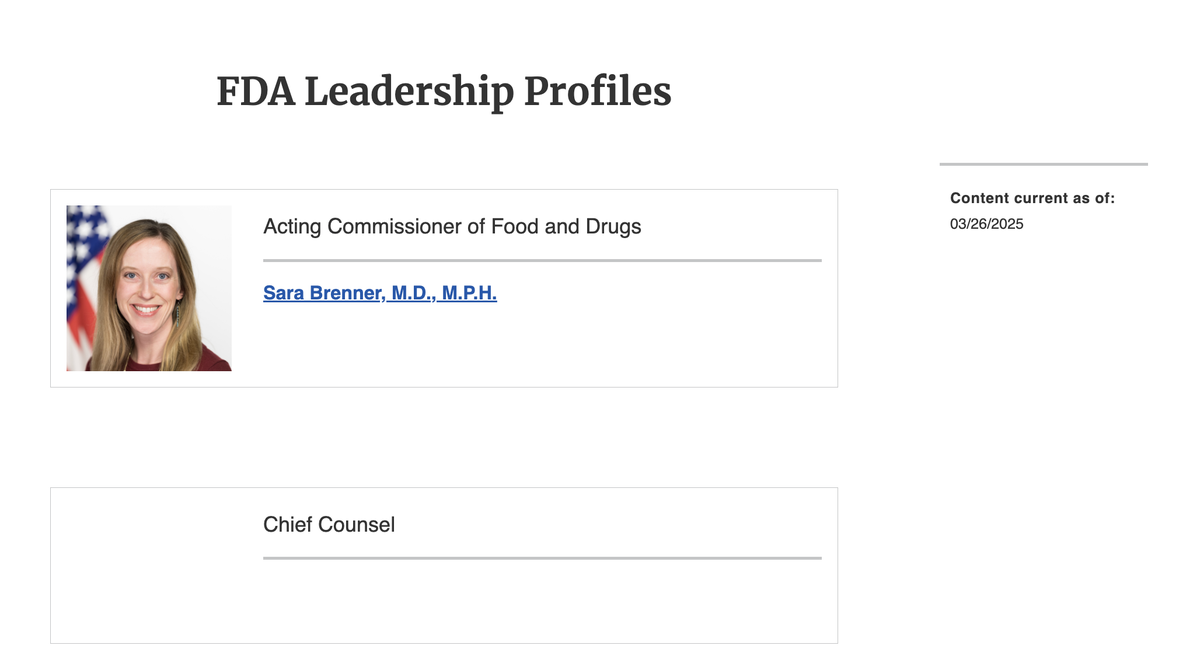
According to a search on the FDA's official website, as of March 26, Sara Brenner is still listed as the acting director of the FDA. However, the FDA's Facebook account has already officially congratulated Makary on his appointment as the 27th director of the FDA.

As per tradition, the Senate's personnel appointment vote is largely divided along party lines, but three Democratic senators crossed party lines to support Makary, indicating that some members of the opposition party are also willing to support his nomination, showing trust in him.
Advocate for collaborating with law enforcement to crack down on illegal e-cigarette activities
During the hearing on March 6, Makary expressed concerns about the large influx of illegally flavored e-cigarette products from China into the US market. He emphasized worries about the unknown ingredients in these products and the difficulty of public health research keeping up with their rapid development. Makary suggested strengthening enforcement against these illegal products, potentially requiring the involvement of the Department of Justice and the FDA's inspection and investigation office.
In the past few years, four new e-cigarette stores have opened near my home. We don't know what these products contain, and even public health officials are unable to research them because studies take a long time to catch up. This is very concerning, and banning them is not the solution.
Makary proposed measures to combat illegal products by utilizing inspection and investigation offices to cooperate with the Ministry of Justice and other law enforcement agencies.
Democratic Senator Dick Durbin, who has been vocal on the issue of e-cigarettes, raised concerns about regulating addiction and harm caused by e-cigarettes during the hearing. He particularly focused on the issue of youth usage. Durbin emphasized that the FDA has failed to enforce the law requiring e-cigarettes to prove they are "appropriate for protecting public health" before entering the market.
We need an FDA Commissioner who is willing to stand up against pharmaceutical and tobacco giants, as well as the President and the Secretary of Health and Human Services, when it comes to protecting the health and well-being of the American people," Durbin’s personal website news briefing stated.
In the end, Durbin demonstrated his trustworthiness by not casting a dissenting vote along party lines, unlike the other two Democrats.

Some individuals believe that Makary is incorrect in placing such a heavy emphasis on anti-smoking policies, and instead should focus on establishing a "scientific consensus around reducing the harm of tobacco," by seeking alternatives to traditional cigarettes and other tobacco products.
A study has confirmed that vaping and HNB technology are much less harmful than traditional cigarettes. Additionally, new products such as nicotine pouches are even less harmful, in some cases equivalent to traditional smoking cessation products like nicotine gum, according to Julio Fuentes, Chairman and CEO of the Florida Hispanic Chamber of Commerce, in his personal column.
Potential regulatory style
The public is more concerned about whether Marty Makary's leadership of the FDA will bring a new atmosphere and change.
The FDA is currently facing pressure to handle the millions of applications for e-cigarette products (PMTAs). Public health organizations criticize the FDA for not acting swiftly and thoroughly enough in rejecting flavored e-cigarette products (especially those popular among teenagers), while the e-cigarette industry complains about unclear approval standards and a lengthy process.
Based on his statements at the hearing, Makary's regulatory style may lean more towards strong enforcement against illegal and non-compliant products. With his high level of concern for illegal e-cigarettes and recommendation for stricter cross-departmental enforcement actions, he may prioritize cracking down on unauthorized products that could potentially pose a threat to public health.
In addition, Makary also stated at the hearing that he will be more open to scientific dissent, emphasizing the role of scientific research and data analysis in decision-making.
While his position on broader tobacco and e-cigarette regulations is still emerging, his tough stance on illegal products and commitment to science indicate that the FDA's future regulation in these areas may prioritize enforcement and evidence-based decision-making.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com









