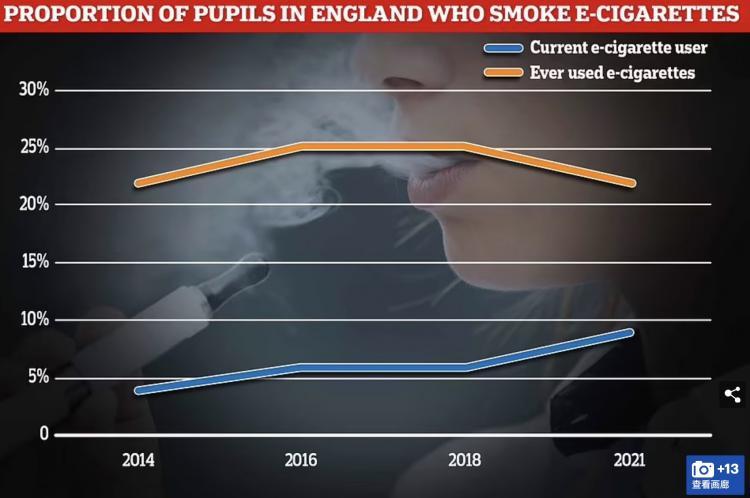
According to official data that indicates the scale of e-cigarette popularity in the country, one in ten secondary school students in England currently use e-cigarettes.
Teenagers are avoiding tobacco and instead opting for trendy electronic devices, which have become the newest must-have accessory to brighten up classrooms. Their popularity has dramatically increased and raised concerns that the UK is slowly sleepwalking into a public health "disaster".
Leading experts expressed concern today over the popularity of these small items (priced at only £4), accusing manufacturers of targeting children with colorful packaging, fruit flavors, and trendy names.
A new call is also demanding the abolition of advertising regulations. The Royal College of Pediatrics and Child Health is calling for strict action to prevent "generations" from becoming addicted to nicotine.
NHS Digital conducted a survey last year on the smoking, drug use, and alcohol habits of nearly 10,000 students between the ages of 11 and 15. The study found that 9% of students currently use e-cigarettes, which is the highest on record since the survey began in 2014.
Girls appear to be driving this trend, with their e-cigarette use rate doubling over the past three years, while boys' e-cigarette use rate has remained steady for the past five years.
Experts have also criticized the rise of social media, on which videos demonstrating vaping techniques have received millions of views. A government-funded report this year found that nearly half of users had watched e-cigarette videos on TikTok, Instagram, or Snapchat. Additionally, children's companies have been targeted for their promotion of vaping products.
Although the popularity of electronic cigarettes has increased, with a 50% growth rate in just three years, the proportion of students smoking traditional cigarettes has remained stable. Currently, one in every 33 students smokes, compared to a peak of one in four during the 1990s.
Statement
This article is compiled from third-party information and is intended for industry exchange and learning purposes only.
This article does not represent the views of 2FIRSTS and 2FIRSTS cannot confirm the authenticity or accuracy of the contents. The translation of this article is only for industry communication and research purposes.
Due to limitations in the translation abilities, the translated article may not completely reflect the original text. Please refer to the original text for accuracy.
2FIRSTS maintains complete alignment with the Chinese government on all domestic, Hong Kong, Macau, and Taiwan-related, and foreign-related statements and positions.
The copyright of the compiled information belongs to the original media and author. If there is any infringement, please contact us for removal.
Disclaimer
This article is provided solely for professional research, industry discussion, and informational purposes. Any references to brands, companies, products, technologies, or policies are made for factual reporting and analytical purposes only, and do not constitute endorsement, recommendation, promotion, or advertising by 2Firsts.
Nicotine-containing products, including but not limited to cigarettes, e-cigarettes, heated tobacco products, and nicotine pouches, carry significant health risks. Readers are responsible for complying with all applicable laws and regulations in their respective jurisdictions, including age restrictions and access limitations.
The information contained in this article should not be regarded as investment, legal, medical, regulatory, or commercial advice. While 2Firsts strives to ensure the accuracy and reliability of its content, it does not assume liability for any direct or indirect loss arising from errors, omissions, inaccuracies, or reliance on the information contained herein.
This article is not intended for individuals below the legal age for accessing tobacco or nicotine-related information in their jurisdiction.
Copyright Notice
This article is either original content produced by 2Firsts or content reproduced, translated, summarized, or adapted from third-party sources with attribution where applicable. The intellectual property rights of the original content remain with 2Firsts or the respective original rights holders.
No individual or organization may copy, reproduce, distribute, republish, modify, translate, or otherwise use this content without prior authorization. Any unauthorized use may result in legal action.
For copyright-related inquiries, corrections, or removal requests, please contact: info@2firsts.com.
AI-Assisted Translation and Editing Notice
Portions of this article may have been translated, edited, or reviewed with the assistance of artificial intelligence tools to improve efficiency and readability. Due to the limitations of AI-assisted translation and editing, discrepancies, omissions, or inaccuracies may exist when compared with the original source.
Where applicable, readers are advised to refer to the original source for the most complete and accurate information. If you identify any errors or believe that any content infringes upon your rights, please contact us at info@2firsts.com, and we will review and address the matter promptly.










