
Key Points (English)
- Two separate EO 12866 meeting requests were filed under RIN 0910-ZC78 within days.
- The draft guidance seeks to clarify evidentiary standards for flavored ENDS under the APPH statutory framework.
- Breeze Smoke reported a 90% revenue decline following intensified FDA enforcement in 2025.
- Glas, represented by a leading FDA regulatory law firm, has also engaged in the White House review process.
- FDA submitted the draft guidance to OIRA on Feb. 18, 2026, initiating interagency review.
- OIRA evaluates regulatory analysis and economic impact but does not override FDA’s scientific determinations.
- The final guidance may further narrow or clarify the authorization pathway for flavored ENDS products.
2Firsts March 3, 2026
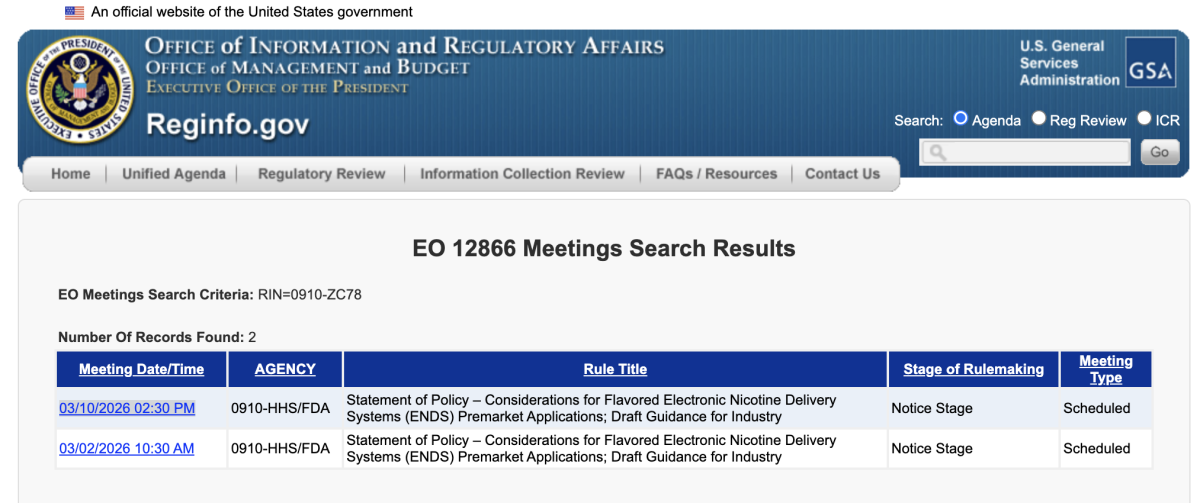
Two separate Executive Order 12866 meetings have been requested under the same regulatory identifier — RIN 0910-ZC78 — signaling heightened industry attention as the U.S. Food and Drug Administration (FDA) advances draft guidance on flavored electronic nicotine delivery systems (ENDS).
According to RegInfo.gov filings, the policy remains at the “Notice” stage. However, parallel White House review requests from different segments of the vaping industry suggest the guidance has become a focal point in the evolving U.S. regulatory landscape.
Two Meetings, Distinct Applicants
Public records show:
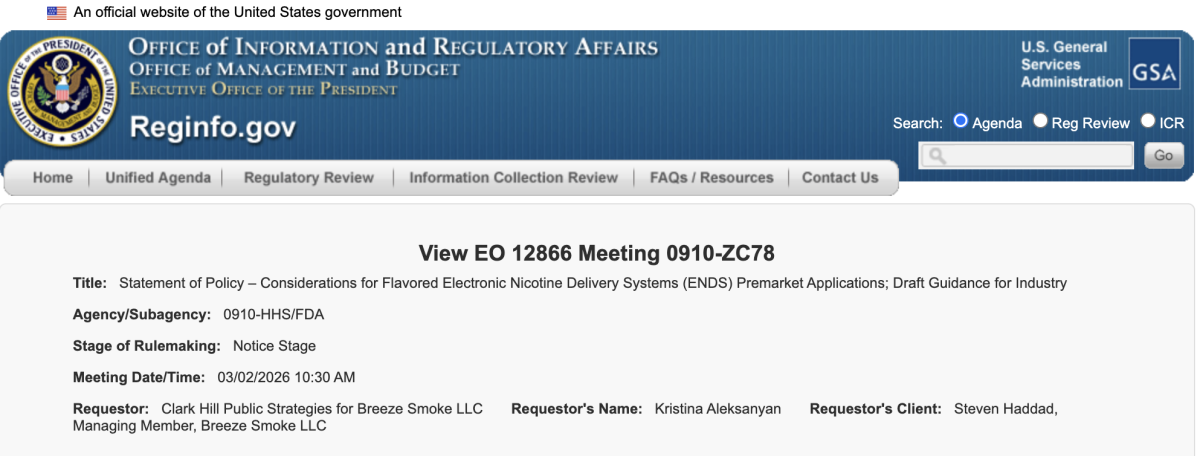
March 2, 2026 – Distributor Breeze Smoke LLC, represented by Clark Hill Public Strategies, requested an EO 12866 meeting (Meeting ID 1324024).
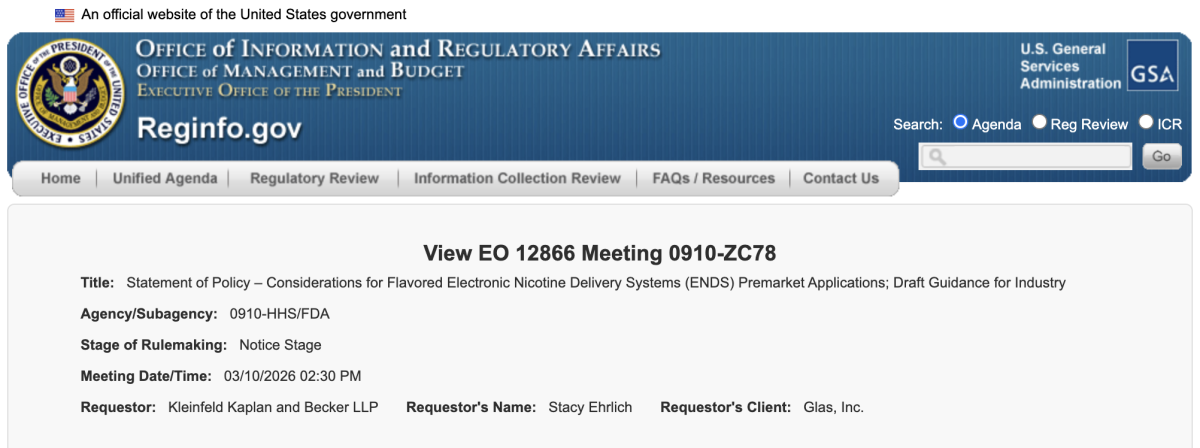
March 10, 2026 (scheduled) – E-liquid manufacturer and brand owner Glas, Inc. filed a separate meeting request (Meeting ID 1329273), represented by Stacy Ehrlich of Kleinfeld, Kaplan and Becker, a law firm widely known for its FDA regulatory practice.
Though filed independently, both meetings concern the same regulatory item, indicating that the draft guidance may affect multiple tiers of the ENDS supply chain — from distribution to product manufacturing.
The Policy at Issue: RIN 0910-ZC78
RIN 0910-ZC78 is titled “Premarket Tobacco Product Applications for Flavored Electronic Nicotine Delivery Systems (ENDS): Draft Guidance.”
As a guidance document rather than a formal rule, it is intended to clarify FDA’s evidentiary expectations when reviewing PMTAs for flavored ENDS products.
Under the Family Smoking Prevention and Tobacco Control Act, FDA must determine that a product is “appropriate for the protection of public health” (APPH) before granting marketing authorization. In prior PMTA decisions, flavored products have faced heightened scrutiny, with the agency requiring product-specific and population-level evidence demonstrating that potential benefits to adult smokers outweigh risks of youth initiation.
Since 2020, FDA has issued Marketing Denial Orders (MDOs) for the vast majority of non-tobacco-flavored ENDS applications, while granting authorization primarily to tobacco-flavored products and a limited number of device platforms.
Economic Pressures and Approval Uncertainty
Breeze Smoke, headquartered in Hazel Park, Michigan, reported approximately $311 million in revenue in 2024, according to Crain’s Detroit Business (Sept. 11, 2025). The publication reported that intensified FDA enforcement actions in 2025 — including import detentions and compliance measures targeting unauthorized ENDS products — led to a revenue decline of roughly 90% by summer 2025.
Breeze products have not received FDA marketing authorization.
Companies with pending PMTA applications are also closely monitoring the draft guidance.
2Firsts previously reported that a suspected backend status update on FDA’s website — later withdrawn — suggested that Glas may be approaching a critical stage in the PMTA review process. While the agency did not confirm the update, the episode drew industry attention to Glas’s regulatory trajectory.

If Glas’s application is indeed nearing final review, clarification of evidentiary standards under RIN 0910-ZC78 could introduce additional variables into its authorization pathway.
Industry observers view Glas’s engagement of Kleinfeld, Kaplan and Becker in the EO 12866 process as part of a broader regulatory risk management strategy aimed at ensuring consistency and predictability in PMTA evaluation standards.
Regulatory Procedure Under EO 12866
Under Executive Order 12866, significant regulatory actions are typically submitted to the Office of Information and Regulatory Affairs (OIRA), within the Executive Office of the President, for interagency review before finalization.
FDA submitted the draft flavored ENDS guidance to OIRA on February 18, 2026, initiating this review phase. During this period, affected stakeholders may request meetings to present views regarding regulatory analysis, economic impact, and policy consistency.
Importantly, guidance documents do not themselves create new legal obligations in the same manner as formal rules. Rather, they articulate the agency’s interpretation and intended application of existing statutory standards.
OIRA’s role is to evaluate regulatory analysis and cross-agency considerations. It does not determine whether a product satisfies the APPH public health standard, nor does it substitute for FDA’s scientific judgment.
A Broader Regulatory Signal
The emergence of multiple EO 12866 meeting requests under a single RIN within days underscores the sensitivity of flavored ENDS policy at the federal level.
Federal regulators continue to emphasize youth prevention as a central objective, while industry participants focus on harm-reduction pathways for adult smokers and the predictability of regulatory standards.
The final language of the guidance may further define the practical authorization boundary for flavored ENDS products in the U.S. market.
What Comes Next
It remains unclear whether OIRA will request additional economic analysis or revisions before the guidance advances.
2Firsts will continue monitoring developments related to RIN 0910-ZC78 and potential implications for the U.S. flavored ENDS market and global supply chains.
(Cover image generated by AI)
For the latest developments in the global vape industry, follow 2Firsts.









