
ecently, the BMJ Group’s authoritative journal Tobacco Control published an article entitled Early assessment of compliance with California’s flavoured tobacco sales prohibition: evidence from vape shops. The study, authored by Arzoo Alam, Lisa Henriksen, Trent O. Johnson, Judith J. Prochaska, and Nina Schleicher, was officially published online in April 2025.
The research focused on California’s flavored tobacco and nicotine product sales ban (SB 793), which took effect on December 21, 2022. California is the second U.S. state after Massachusetts to implement a comprehensive prohibition on flavored tobacco sales. The study aimed to assess compliance in the early months following the ban, particularly whether vape shops continued to illegally sell flavored products.
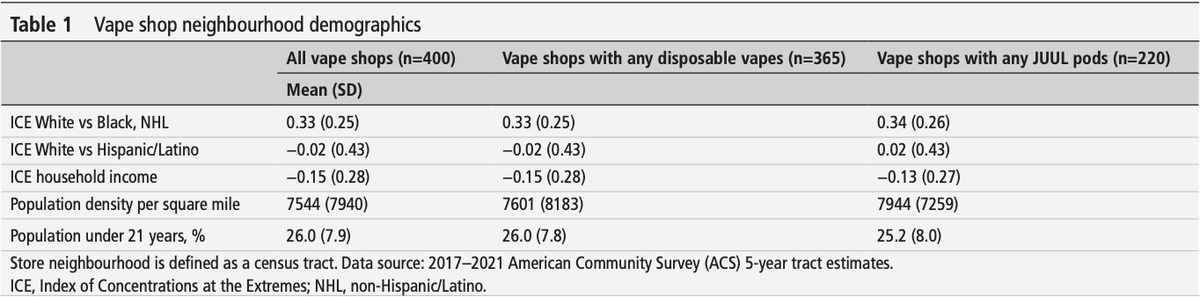
Between April and July 2023, field observations were conducted at 400 vape shops in California, selected from a 2019 sample of retailers confirmed to still be operating. Researchers documented the actual availability of disposable nicotine e-cigarettes, JUUL pods, and their flavored versions (limited to in-stock items). Shops were grouped by proximity to university campuses (≤3 miles vs. >3 miles) and by community demographics (race, income, proportion of young residents). Logistic regression analysis was used to examine associations between flavored product sales and geographic or demographic characteristics.

Key Findings
High noncompliance rate
- 50.3% of vape shops continued to sell flavored disposable e-cigarettes or JUUL pods.
- Among shops selling disposable e-cigarettes, 53.2% offered flavored versions.
- Among shops selling JUUL pods, 30.9% offered flavored versions.
No significant correlation with campus proximity or demographics
- Shops near universities (≤3 miles) had a 48.7% violation rate, compared with 51.7% for those farther away (>3 miles), with no significant difference.
- No statistically significant differences were observed across communities with different population densities, proportions of residents under 21, racial/ethnic composition (White vs. Black/Hispanic), or income levels.
Possible reasons for violations
- Retailers clearing inventory immediately after the ban.
- Lack of awareness or understanding of the law.
- Weak enforcement, with fines (USD 250) too low to act as a deterrent.
Conclusions and Recommendations
Limited early impact: Four to seven months after the law took effect, the violation rate remained around 50%, with no evidence of improvement over time.
Need for stronger enforcement: Recommendations include increasing fine amounts and implementing license revocation. California enacted AB 935 in January 2024, providing for license revocation after three violations and authorizing public health departments to directly participate in enforcement.
Education and outreach: Experiences in San Francisco and Minneapolis demonstrate that face-to-face education and regular inspections can significantly improve compliance.
In summary, the study concluded that California’s flavored tobacco ban faced serious enforcement challenges in its early phase. To achieve real effectiveness, stricter enforcement, stronger penalties, and broader retailer education are required.
Study Limitations
- Focused only on vape shops, excluding other tobacco retailers.
- Did not systematically investigate “concept flavors” (products with vague names to evade the ban), potentially underestimating violation rates.
- Did not use “purchase testing,” so some covert sales may not have been captured.
Article Information
Title
Early assessment of compliance with California’s flavoured tobacco sales prohibition: evidence from vape shops
Authors
Arzoo Alam, Lisa Henriksen, Trent O. Johnson, Judith J. Prochaska, Nina Schleicher
Corresponding Author (Author note)
Arzoo Alam, MPH
Stanford University School of Medicine, Prevention Research Center
3180 Porter Drive, Palo Alto, CA, USA
Email: arzoo.alam@stanford.edu
Publication Date
April 2025
Journal
Tobacco Control (BMJ Publishing Group)
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










