
Special statement:
This article is for internal industry communication only and does not make any recommendations for specific brands or products.
The images presented in this article are only used to describe the facts and are not intended as advertisements for any products.
Minors are prohibited from accessing this article.
Product Highlights:
·Large Capacity: The "2+10" design provides a total e-liquid capacity of 12ml and up to 5500 puffs.
·Extended Battery Life: The built-in 1000mAh battery supports prolonged use.
·Regulatory Compliance: The product was officially listed by the UK Medicines and Healthcare products Regulatory Agency (MHRA) in May, ensuring it meets local market regulations.
·Compliant Alternative: The ELFBAR AF5500 is likely a compliant replacement for the ELFBAR AF5000 e-cigarette.
September 4, 2025 - Recently, 2Firsts has noted that a new e-cigarette product from the iMiracle brand ELFBAR, the "ELFBAR AF5500," has officially appeared on major UK e-cigarette distributor websites after being publicly listed by the UK Medicines and Healthcare products Regulatory Agency (MHRA).
"2+10" Design with 5500 Puffs
2Firsts has collected and compiled the relevant specifications for the ELFBAR AF5500, as shown in the chart below:
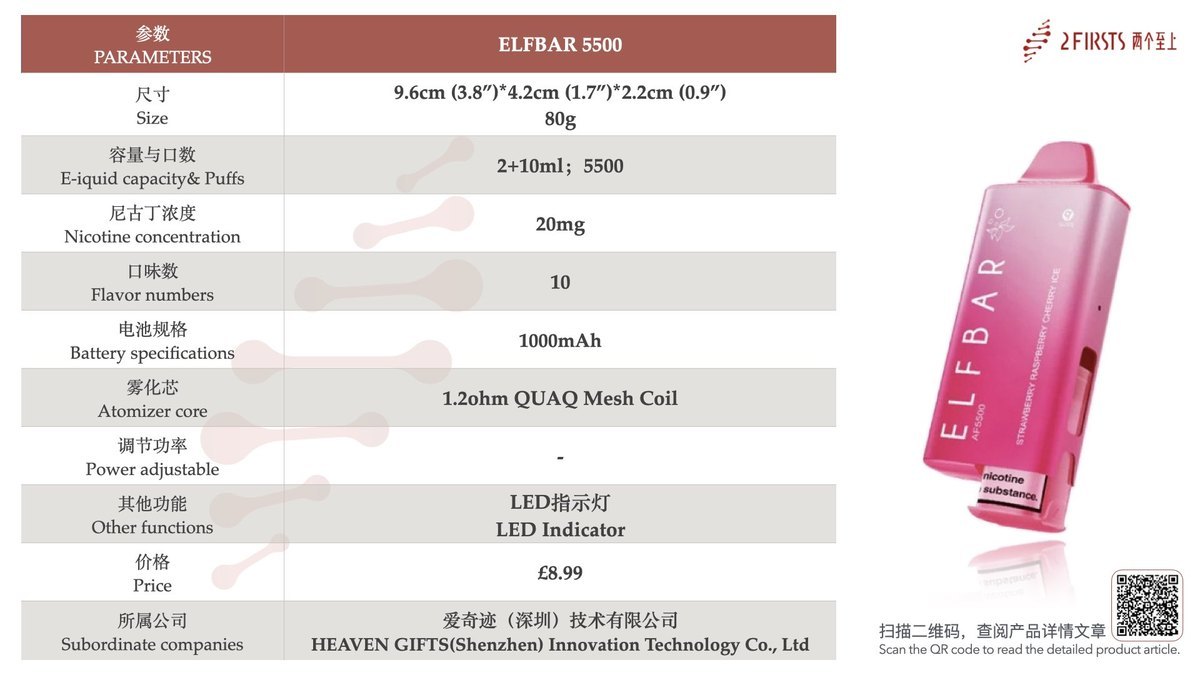
Based on the specifications, the ELFBAR AF5500 e-cigarette features a "2+10" design, a nicotine concentration of 20mg, a total e-liquid capacity of 12ml, and approximately 5500 puffs. This works out to about 458 puffs per milliliter of e-liquid.

The ELFBAR AF5500 has a 1000mAh battery capacity and is currently available in 10 flavors, primarily focusing on "mixed fruit" and "fruit + ice" options.
Likely a Compliant Alternative to the ELFBAR AF5000
Following the implementation of the UK's disposable vape ban in June of this year, 2Firsts observed that the ELFBAR AF5000—one of the "2+10" products that was a bestseller in the region—was taken down from many major e-commerce platforms or marked as discontinued. At the same time, the official ELFBAR UK website indicated that a replacement pod version of the AF5000 was set to be launched.
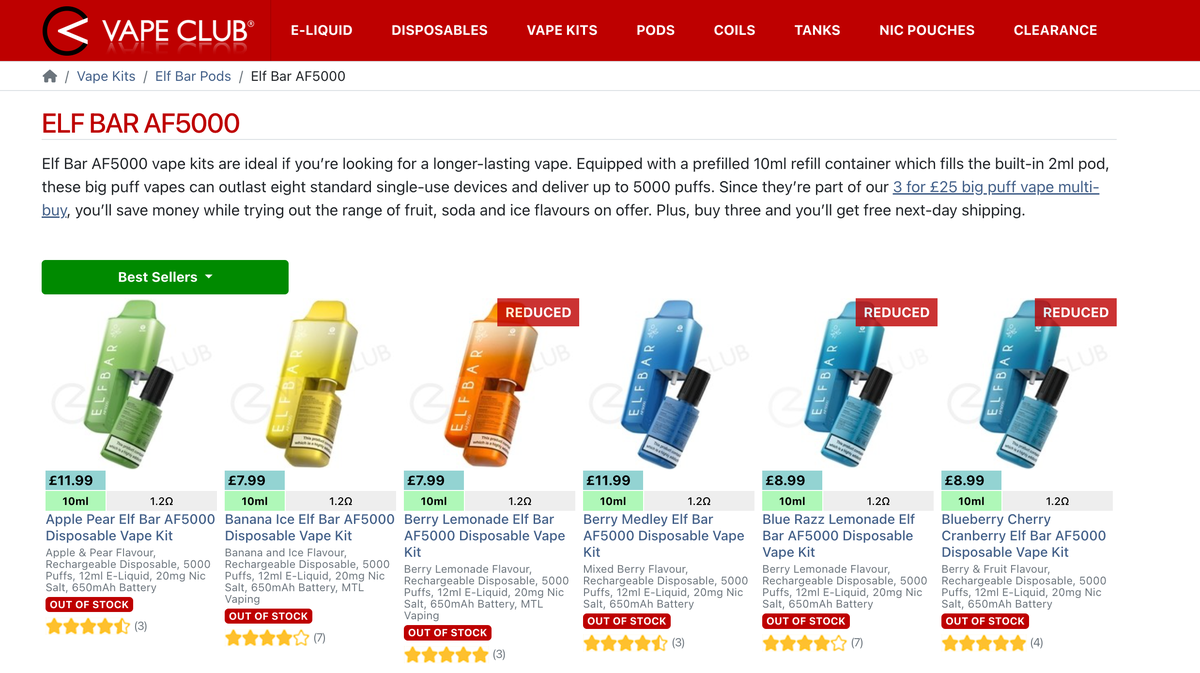
According to the distributor website Vape Sourcing, the ELFBAR AF5500 kit has a similar design to the ELFBAR AF5000 but is actually an upgraded reusable e-cigarette.

Therefore, the ELFBAR AF5500 is likely the compliant alternative to the ELFBAR AF5000 e-cigarette.
Publicly Listed on MHRA, Priced at £8.99
Furthermore, a search of the UK Medicines and Healthcare products Regulatory Agency's (MHRA) e-cigarette product notification database by 2Firsts revealed that the "ELFBAR AF5000 Device 1000 mAh" and "ELFBAR AF5500 Device 1000 mAh" were applied for and publicly listed as a single SKU by HEAVEN GIFTS GERMANY on May 27, 2025.

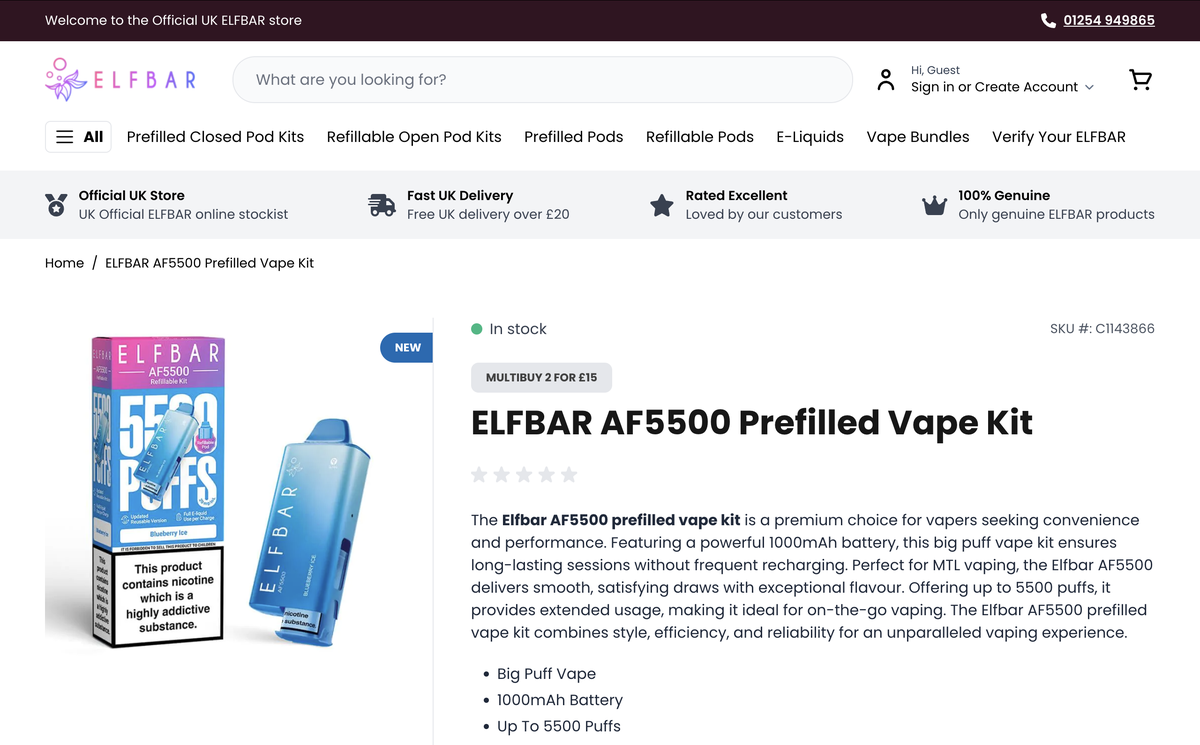
Currently, the ELFBAR AF5500 e-cigarette is available on UK e-cigarette retail websites such as ELFBAR.UK, Vape Sourcing, and Vape Club, with a product price of approximately £8.99.
As a leading global NGP media and think tank, 2Firsts is dedicated to providing the latest product and technological information and insights to practitioners around the world in various categories such as e-cigarettes, heated tobacco products, and modern oral products. It aims to drive technological changes and innovations in NGP products worldwide, thereby offering tobacco consumers globally with safer products and lifestyles.
With a source of information covering the supply chains in China and global markets, 2Firsts product coverage has become one of the most influential platforms for new product and technology releases globally.
Contact 2Firsts for the following services:
1. Providing leads on new products and technologies;
2. Offering comments on products and technologies;
3. Seeking media coverage for your products;
4. Identifying sales channels for products.
Contact Information: Email: info@2firsts.com
Contact CEO Alan Zhao of 2Firsts on LinkedIn.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com









