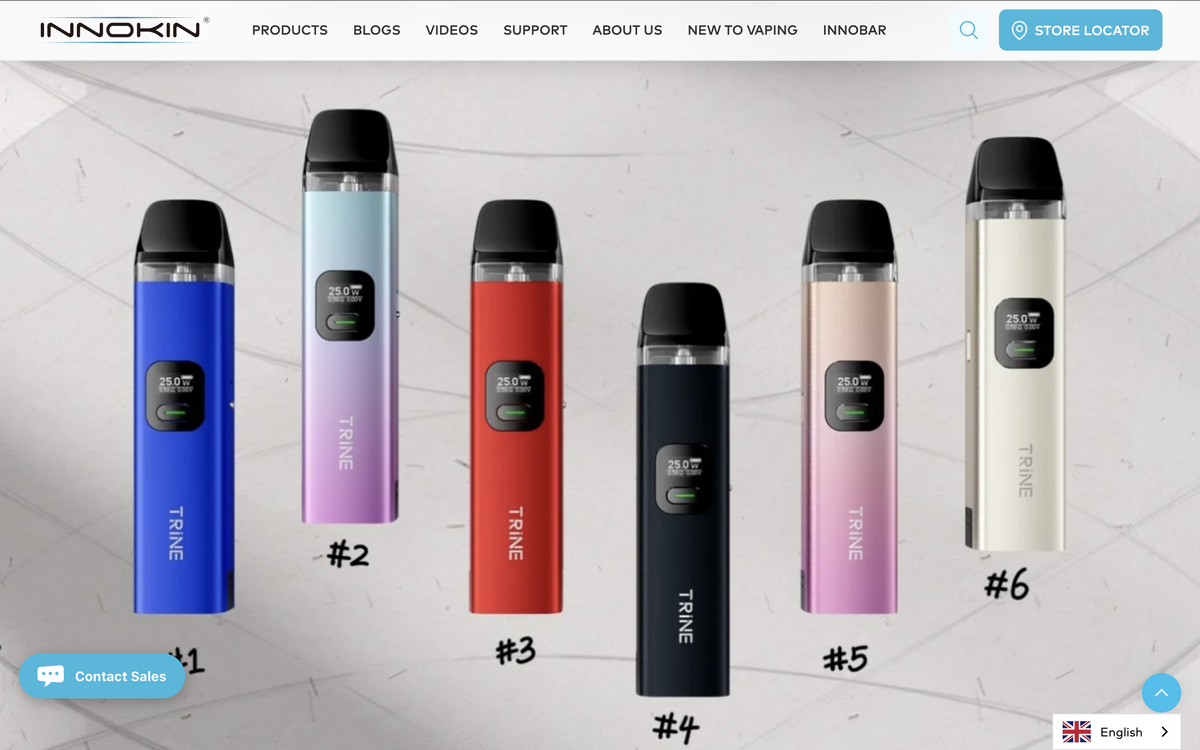
Recently, 2Firsts noticed a new type of replaceable battery e-cigarette called the Innokin Trine being showcased at a tobacco exhibition in Dortmund. The product, launched by e-cigarette brand Innokin, is now available on e-cigarette retail websites in regions such as the UK and Germany. With its design allowing for replaceable batteries, the product complies with the new regulations set to be implemented by the EU Battery Directive in 2026.

At the recent Dortmund tobacco exhibition, several exhibitors showcased e-cigarette products with replaceable batteries, including disposable, pod-based, and open-system devices, in response to the EU's Battery Directive. According to the directive, by 2026, batteries in portable devices (such as disposable e-cigarettes) must be easily removable and replaceable by the user.

The newly released Innokin Trine, an open system product from Innokin, complies with the upcoming new regulations in the European Union market.

In addition, the main feature of Trine is its "three-in-one" structure: atomizer, detachable and rechargeable battery, control panel. At the same time, the detachable and rechargeable battery enhances the convenience and durability of the device.

Specifically, the main product features of the Innokin Trine include:
E-liquid capacity: 2ml Nicotine concentration: Supports a variety of nicotine levels in e-liquid Battery capacity: 1000mAh removable battery Charging method: Supports Type-C fast charging.
Trine Power Hub charger charges
Backup batteries can also be purchased separately.
Power Mode: Provides a power output of 6-25W.
Function: - Suction activation
Support mouth to lung (MTL) or limit direct lung (RDL) inhale mode.
Compatible with a variety of e-liquid flavors.
Charging indicator light
The screen displays operational parameters such as output power and battery level.
Currently, the Innokin Trine has been listed on the official website of the Innokin e-cigarette brand and major distributor websites in Europe, with a selling price of approximately 19.95 euros.
In addition to the Innokin brand, other brands such as GOLISI and NEXIONE have also introduced similar products with replaceable batteries. 2Firsts will continue to monitor the latest products and market trends in replaceable battery e-cigarette products, providing the public with accurate and authoritative industry information.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










