
The organizers of the Spanish tobacco expo EVO NXT held a forum at the event focused on the analysis of the EU nicotine pouch market. During the forum, Jonas Lundqvist, CEO of the Northern European Nicotine Pouches Alliance (NNPA.EU), shared insights on the EU nicotine pouch market.
According to Jonas, there are over 90 nicotine pouch brands in the EU's 27 countries, with physical retail accounting for 68% and online retail accounting for 32%. This includes both brick-and-mortar stores and online platforms. There are over 100,000 total sales points for nicotine pouches, including 104,000 physical retail points and 90 online retail points.
The manufacturing of nicotine pouches is mainly concentrated in countries such as Sweden, Poland, Italy, and Czech Republic. Despite the currently undeveloped market not showing high activity, due to high smoking rates and large populations, it is expected that these regions will become important markets for nicotine pouches in the future.
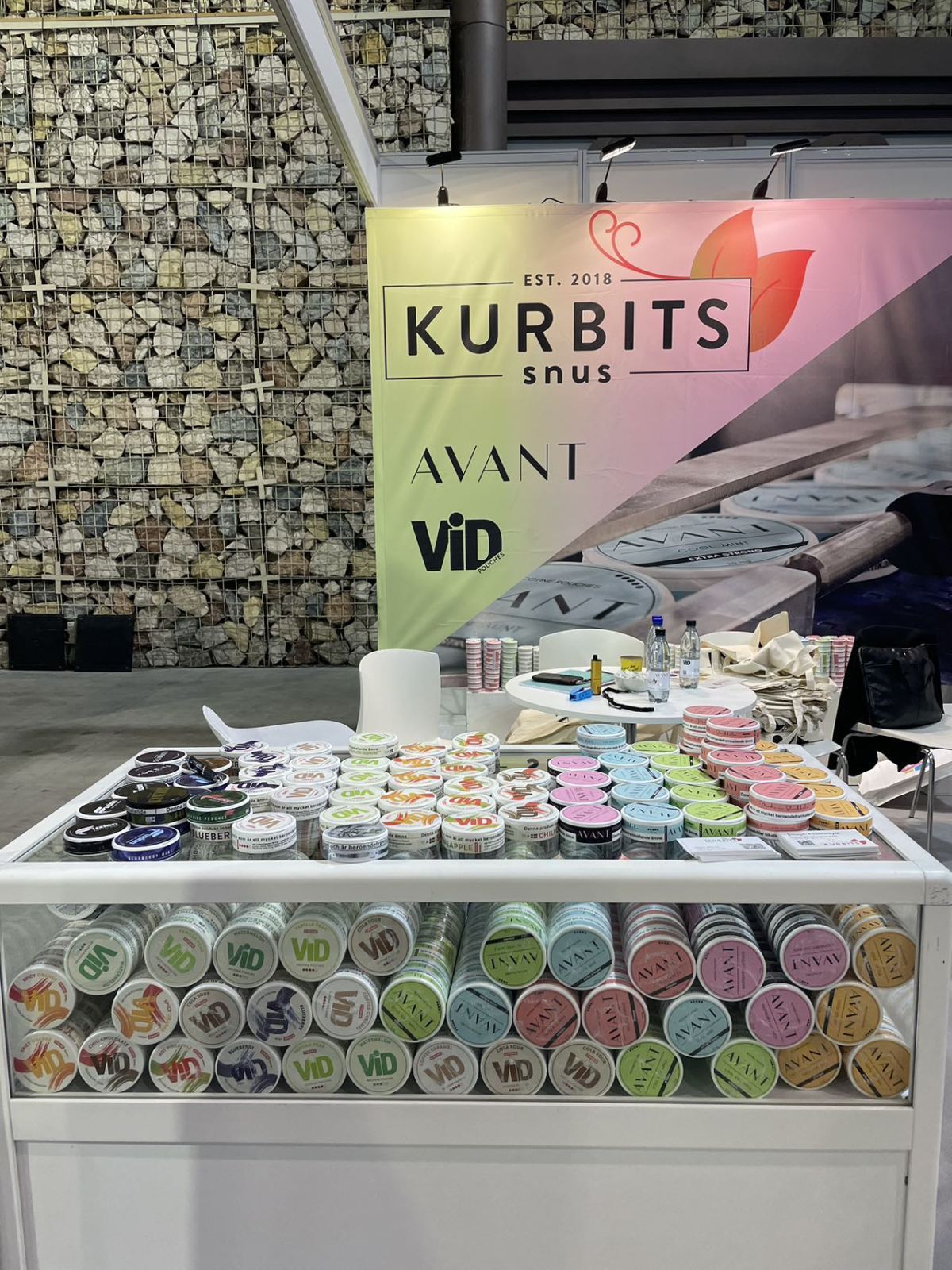
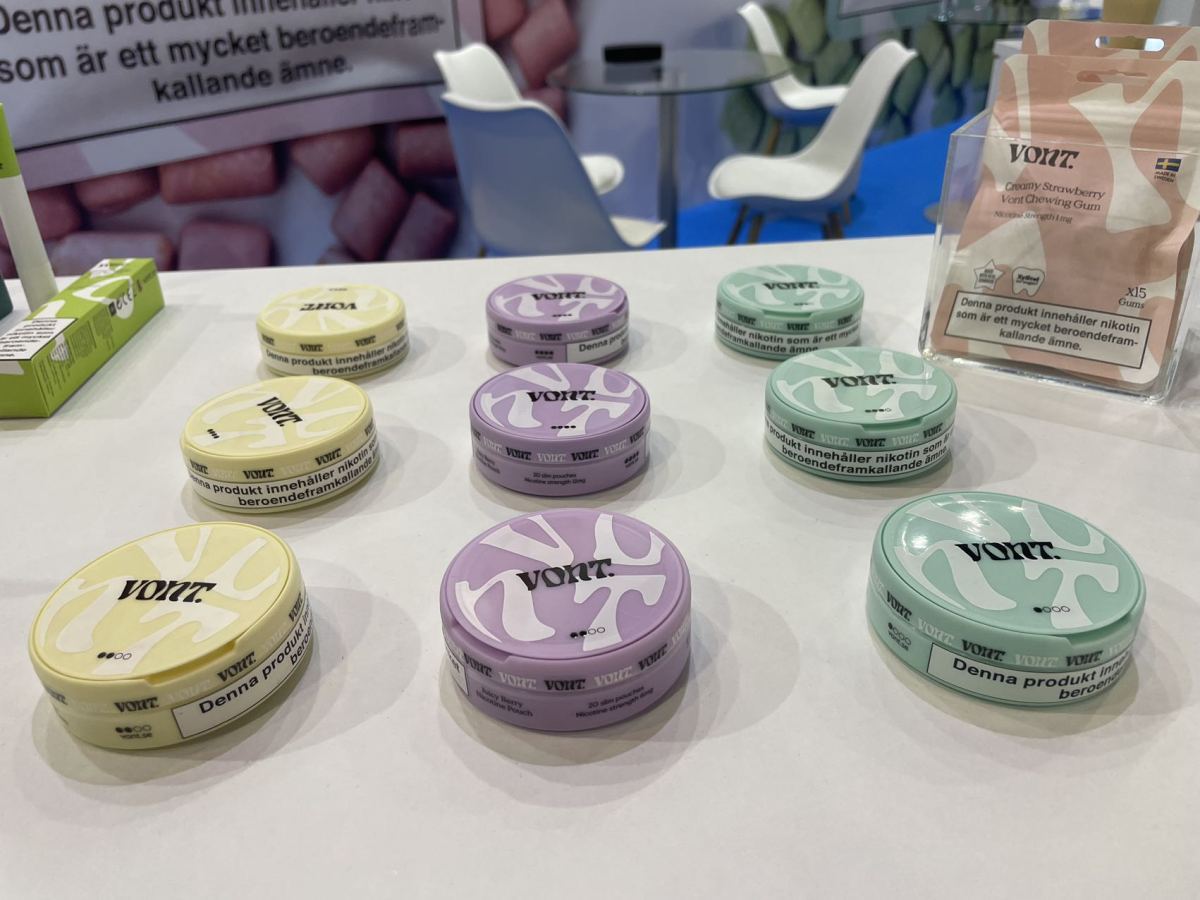
2FIRSTS surveyed the brands that introduced nicotine pouch products at the EVO NXT exhibition.





We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










