The regulation of electronic cigarettes is becoming stricter in the EU. IEVA is calling for increased communication between Chinese companies and the EU authorities.
On September 29th, 2022, Shenzhen-based company 2FIRSTS Technology Limited (www.2firsts.com) was invited by the Independent European Vape Alliance (IEVA) to attend and participate in the IEVA 2022 conference in Brussels, Belgium. The conference provided up-to-date information on the dynamic trends and regulations in the electronic cigarette market in the European Union and its member countries, clarifying the current state of the industry and predicting future trends. IEVA President Dustin Dahlmann chaired the meeting.
At the 2022 IEVA conference in Brussels, as seen in image source 2 from Firsts.
2FIRSTS invited to join IEVA as its member. Dustin Dahlmann announced the news during the meeting and welcomed 2FIRSTS' membership.
In early 2022, 2FIRSTS applied to join the Electronic Cigarette Professional Committee (ECCC) of the China Electronic Commerce Association and became a director unit. Additionally, 2FIRSTS is a global industry media outlet for electronic cigarettes. By joining the IEVA, 2FIRSTS will engage in comprehensive and in-depth cooperation, connecting Chinese e-cigarette companies with the European Union market, thus promoting the integration and communication of regulatory information between the Chinese and European e-cigarette markets and supporting Chinese e-cigarette companies in going global, while achieving mutual benefits for the e-cigarette industry in China and Europe.
The EU electronic cigarette regulation was discussed in three agenda items during the meeting.
Looking back at key decisions made by the EU regarding regulation of e-cigarettes, such as the TPD, Battery Regulation, Directive on reducing packaging waste, etc.
Representatives from various countries provided an update on their respective regulations and market trends regarding electronic cigarettes.
Reaffirming the member responsibility guidelines and introducing additional regulations in member management.
During a discussion on the future trends of the industry, Dustin Dahlmann, president of IEVA, gave a keynote speech on the main trends and challenges in the electronic cigarette industry. Representatives from various countries then shared the latest developments in their respective national electronic cigarette regulatory policies. During this segment, Jason, the diplomatic assistant of ECCC and international public relations manager of 2FIRSTS, presented the latest developments in electronic cigarette regulation in China.
The regulatory trends in the electronic cigarette industry of the EU were conveyed at the conference through speeches by Dustin Dahlmann and representatives from various countries.
The EU will confirm electronic cigarette consumption tax revenues soon. The current TPD2 in the EU does not provide regulations on electronic cigarette consumption taxes. However, the EU is set to introduce a bill on the modification of electronic cigarette taxes on December 7th, 2022. While more details have yet to be revealed, according to the EU legislative process, this tax bill will only become national law and officially take effect between 2024 and 2026. It is reported that if the aim of tax exemption for electronic cigarettes is not achieved this time, various electronic cigarette associations and related companies in the EU will strive to set the electronic cigarette tax at 0.1 euro per milliliter.
The content of the speech on electronic cigarette taxation, image source: 2FIRSTS.
Countries are establishing relevant taxes for e-cigarette products. For example, Belgium is drafting a tax for the recycling of e-cigarette packaging and batteries, while Germany's e-cigarette tax is set to reach 0.32 euros per milliliter by 2026, significantly higher than the average e-cigarette tax level in the European Union. As a result, the e-cigarette industry in Germany is facing taxation pressure, leading to a decrease in both online and physical e-cigarette stores. Meanwhile, Spain is currently drafting a bill to increase its e-cigarette tax, which is expected to be higher than Germany's e-cigarette tax in 2026.
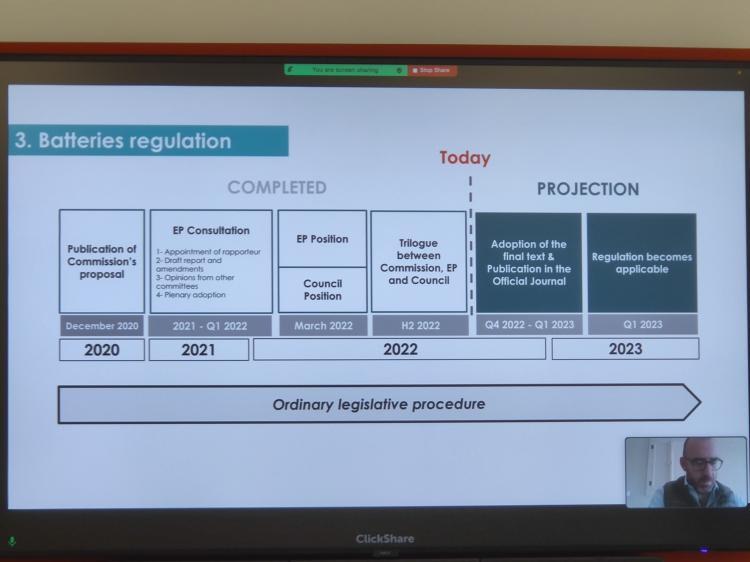
Environmental protection" to become important direction of EU regulation. In addition to tax policies, "environmental protection" has also become an important focus and topic in the EU's policy making. According to the content of the meeting, a battery law for all electronic products is being drafted, which will be finalized and announced in the first quarter of 2023. At that time, all electronic product categories, including the batteries of all electronic cigarette products, must be rechargeable or have replaceable batteries.
Regarding the speech content on e-cigarette batteries, source 2 FIRSTS.
Electronic cigarette product packaging, which is also seen as an environmental indicator, is currently being addressed in relevant legislation. The European Union Commission is set to propose a review of the Packaging and Packaging Waste Directive on November 30, 2022. The review is expected to consider whether the packaging is reusable or recyclable, as well as reducing excessive packaging.
The Brussels Conference conveyed a signal of increasing regulation for e-cigarettes in the EU, from the recycling of batteries and packaging under EU TPD3 regulations, to countries planning their own e-cigarette taxes. Dustin Dahlmann stated that in this context, it is necessary to strengthen communication with Chinese companies, as only with their cooperation can legal compliance be truly implemented.
At the end of the meeting, Dustin Dahlmann reiterated the members' code of conduct and the attending members voted in favor of the newly added regulations for member management.
2FIRSTS will continue to monitor and report on the regulatory policies regarding e-cigarettes in the European Union. For more information on EU e-cigarette regulations, please visit the official 2FIRSTS website: cn.2firsts.com (Chinese) or www.2firsts.com (English).
Statement:
1. This article is intended for internal communication and discussion within the industry. The information in this article has not been confirmed by the European Union. Please refer to official information released by the EU if there is any disagreement. 2. Smoking is harmful to health. Minors are prohibited from reading this article.
This article is an original creation of 2FIRSTS Technology Co., Ltd. in Shenzhen, and its copyright and usage rights belong to the company. Without authorization, no individual or organization shall reproduce, reprint, or use any other means to infringe on the company's copyright. The company will reserve the right to pursue legal responsibility against any violators.
Disclaimer
This article is provided solely for professional research, industry discussion, and informational purposes. Any references to brands, companies, products, technologies, or policies are made for factual reporting and analytical purposes only, and do not constitute endorsement, recommendation, promotion, or advertising by 2Firsts.
Nicotine-containing products, including but not limited to cigarettes, e-cigarettes, heated tobacco products, and nicotine pouches, carry significant health risks. Readers are responsible for complying with all applicable laws and regulations in their respective jurisdictions, including age restrictions and access limitations.
The information contained in this article should not be regarded as investment, legal, medical, regulatory, or commercial advice. While 2Firsts strives to ensure the accuracy and reliability of its content, it does not assume liability for any direct or indirect loss arising from errors, omissions, inaccuracies, or reliance on the information contained herein.
This article is not intended for individuals below the legal age for accessing tobacco or nicotine-related information in their jurisdiction.
Copyright Notice
This article is either original content produced by 2Firsts or content reproduced, translated, summarized, or adapted from third-party sources with attribution where applicable. The intellectual property rights of the original content remain with 2Firsts or the respective original rights holders.
No individual or organization may copy, reproduce, distribute, republish, modify, translate, or otherwise use this content without prior authorization. Any unauthorized use may result in legal action.
For copyright-related inquiries, corrections, or removal requests, please contact: info@2firsts.com.
AI-Assisted Translation and Editing Notice
Portions of this article may have been translated, edited, or reviewed with the assistance of artificial intelligence tools to improve efficiency and readability. Due to the limitations of AI-assisted translation and editing, discrepancies, omissions, or inaccuracies may exist when compared with the original source.
Where applicable, readers are advised to refer to the original source for the most complete and accurate information. If you identify any errors or believe that any content infringes upon your rights, please contact us at info@2firsts.com, and we will review and address the matter promptly.










