
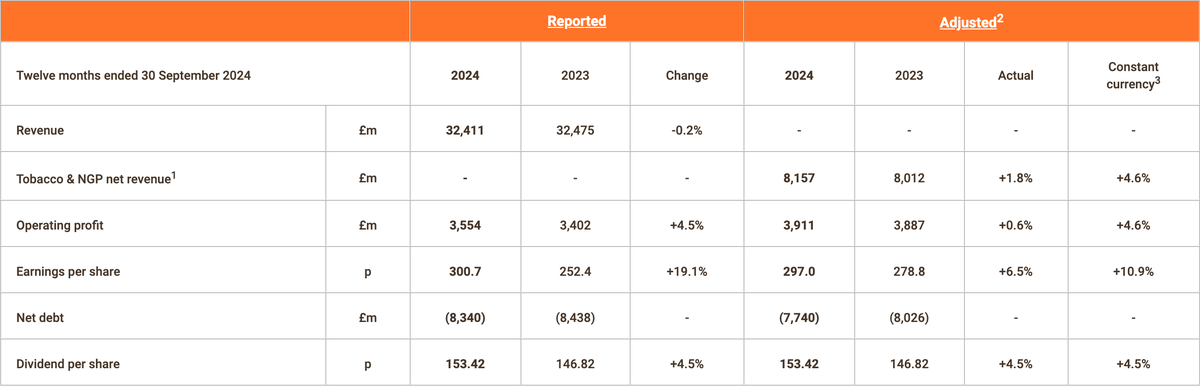
On November 19, Imperial Brands (IMB) announced its full-year financial results for the period ending September 30, 2024. The company reported a slight decline in total revenue, which fell by 0.2% year-on-year to £32.411 billion.
Despite the dip in revenue, adjusted operating profit rose by 4.5% to £3.887 billion. Adjusted net revenue for the "Tobacco and Next Generation Products (NGP)" segment increased by 4.6%, reaching £8.157 billion.
Key highlights of the report:
- Tobacco and NGP net revenue increased by 4.6%.
- Market share gains were reported in four out of IMB’s five priority markets, with an overall increase of 0.05%.
- NGP's net revenue grew by 26%, with all three regions achieving growth and improved margins.
- Logista's growth reflected strong tobacco pricing and benefits from last year’s acquisitions.
- Adjusted earnings per share (EPS) rose by 10.9%, driven by profit growth and a reduced share count, while reported EPS increased by 19.1%.
- Strong cash generation, with free cash flow reaching £2.4 billion.
- IMB plans to return approximately £2.8 billion to shareholders in FY25, including a £1.25 billion share buyback and a 4.5% increase in the FY24 dividend.
The report highlights that overall market share in IMB’s five priority markets increased by 5 basis points (bps). Growth was recorded in the U.S., Germany, Spain, and Australia, while the U.K. saw a decline.
In Europe, strong growth in tobacco and Next Generation Product (NGP) net revenue was noted, with a stable market share in Germany. In the Americas, the successful launch of the modern oral nicotine brand "Zone" drove a 29.4% increase in NGP net revenue. In Africa, Asia, and the Middle East, effective tobacco pricing strategies contributed to further market share expansion.
Notably, NGP's net revenue grew by 26.4%, with all three categories—heated tobacco, vaping, and modern oral —experiencing growth.
Stefan Bomhard, CEO of IMB, said:
"As we enter the final year of our current strategy, the investment we have made in consumer capabilities, cultural transformation and agile ways of working has supported another year of accelerated financial delivery and growing capital returns. "
"In tobacco, investment in our brands and sales force initiatives have delivered aggregate market share gains across our five priority markets, while delivering strong pricing. This was supported by an encouraging stabilisation in German market share for the first time under our strategy"
"In next generation products (NGP), we continue to build scale across our footprint with net revenues up 26.4% at constant currency driven by growth from all three regions and market share growth in all three categories. Our partnership approach to product innovation has enabled us to launch new products across all three categories during the year. This included our successful entry to the fast-growing modern oral category in the US with our brand ‘Zone’."
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










