According to 2FIRSTS at the RELX YUEKE e-cigarette physical store in Indonesia, on October 26th Jakarta time, RELX YUEKE officially launched a new product, the RELX Infinity Plus, for sale both online and offline. The new product has a retail price of IDR 300,000, approximately RMB 138, and is claimed to charge up to 80% in just 30 minutes. It comes in five colors: Black Phantom, Lunar Dust, Hidden Pearl, Rising Tide, and Pink Whisper.
The RELX Infinity Plus is now available in five different colors.
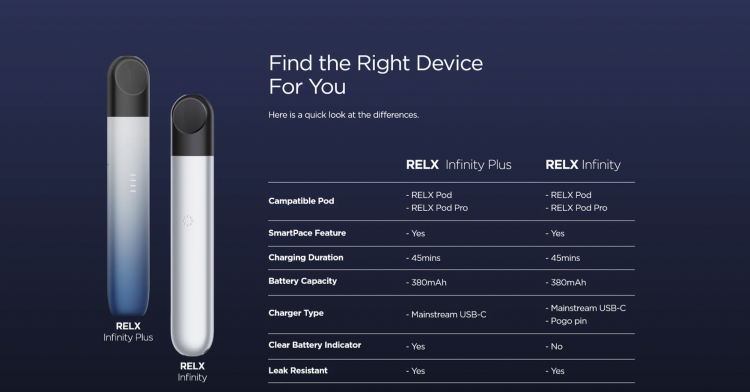
The new product from RELX Yuetheke can now be seen on the official website's homepage. Compared to the old RELX Infinity, it features a clean battery indicator. The standout feature of this new product is its 380mAh battery, which has four levels of remaining power reminders at 25%, 50%, 75% and 100%, providing users with a clearer understanding of their device's battery life. It also has 11 layers of structure to prevent internal leakage and condensation. Additionally, the product can be placed upright, thanks to its flat bottom design, which makes the device easier to stand.
The official website homepage displays that RELX Infinity Plus is releasing new product information, with details about the new product.
Product details, product details, product details.
1. This article is intended solely for internal industry communication and exploration, and does not serve as any form of advertising or recommendation for brands or products. 2. Smoking is harmful to health. Minors are prohibited from reading this article.
This article is an original creation of Shenzhen 2FIRSTS Technology Co., Ltd. The copyright and license to use belong to the company, and any unauthorized copying, reproduction or other infringement of the company's copyright is strictly prohibited. The company reserves the right to pursue legal action against any individual or entity that violates this policy.
Disclaimer
This article is provided solely for professional research, industry discussion, and informational purposes. Any references to brands, companies, products, technologies, or policies are made for factual reporting and analytical purposes only, and do not constitute endorsement, recommendation, promotion, or advertising by 2Firsts.
Nicotine-containing products, including but not limited to cigarettes, e-cigarettes, heated tobacco products, and nicotine pouches, carry significant health risks. Readers are responsible for complying with all applicable laws and regulations in their respective jurisdictions, including age restrictions and access limitations.
The information contained in this article should not be regarded as investment, legal, medical, regulatory, or commercial advice. While 2Firsts strives to ensure the accuracy and reliability of its content, it does not assume liability for any direct or indirect loss arising from errors, omissions, inaccuracies, or reliance on the information contained herein.
This article is not intended for individuals below the legal age for accessing tobacco or nicotine-related information in their jurisdiction.
Copyright Notice
This article is either original content produced by 2Firsts or content reproduced, translated, summarized, or adapted from third-party sources with attribution where applicable. The intellectual property rights of the original content remain with 2Firsts or the respective original rights holders.
No individual or organization may copy, reproduce, distribute, republish, modify, translate, or otherwise use this content without prior authorization. Any unauthorized use may result in legal action.
For copyright-related inquiries, corrections, or removal requests, please contact: info@2firsts.com.
AI-Assisted Translation and Editing Notice
Portions of this article may have been translated, edited, or reviewed with the assistance of artificial intelligence tools to improve efficiency and readability. Due to the limitations of AI-assisted translation and editing, discrepancies, omissions, or inaccuracies may exist when compared with the original source.
Where applicable, readers are advised to refer to the original source for the most complete and accurate information. If you identify any errors or believe that any content infringes upon your rights, please contact us at info@2firsts.com, and we will review and address the matter promptly.










