
The U.S. Food and Drug Administration (FDA) and U.S. Customs and Border Protection (CBP) seized 3 million e-cigarette products with an estimated retail value of $76 million. This is the largest seizure by dollar value in FDA history. To understand the event fully, 2Firsts looked at reports from many U.S. media outlets and public feedback.
US media: FDA exaggerates vaping risks, plans more enforcement actions
Filter Magazine commented on the incident, noting that the FDA's announcement claimed that seizing unapproved e-cigarettes could reduce youth access. However, the magazine criticised the FDA's statement as misleading, suggesting that it exaggerated the potential impact of nicotine on adolescent brain development up to the age of 25 and implied that nicotine use could lead to addiction to other substances.

Filter Magazine also emphasises that despite inadequate regulatory measures, products such as disposable e-cigarettes have played a positive role in reducing smokers' health risks, a trend supported by industry data.

Investors Hangout also reported on the incident, highlighting that the authorities are prepared to take strong legal action against the illegal sale of e-cigarettes, including civil fines and possible criminal convictions, to protect consumers - especially young people - from the dangers of unsafe products.

The Investors Hangout also speculated on future collaboration between the FDA and CBP, predicting continued enforcement actions aimed at ensuring consumer safety and cracking down on the illegal distribution of e-cigarettes.
Social media users: E-cigarette seizures seen as protecting big tobacco interests
On social media platform X (formerly Twitter), international users commented on the FDA's press release, with many expressing criticism and sarcasm towards the FDA and CBP's actions. Concerns were raised about the influence of big tobacco and pharmaceutical companies.
Translation: "Among non-big tobacco e-cigarette products, $76 million was seized. FDA Commissioner Robert Califf said: "These superior e-cigarette products often end up in the hands of smokers, effectively helping them quit deadly tobacco. The danger here is that it could put tobacco control officials like me out of a job.
Think of the smokers who have died because of this. People are dying from FDA-approved tobacco products, yet they won't allow anyone to use anything else unless it's their own deadly product or from Big Pharma.
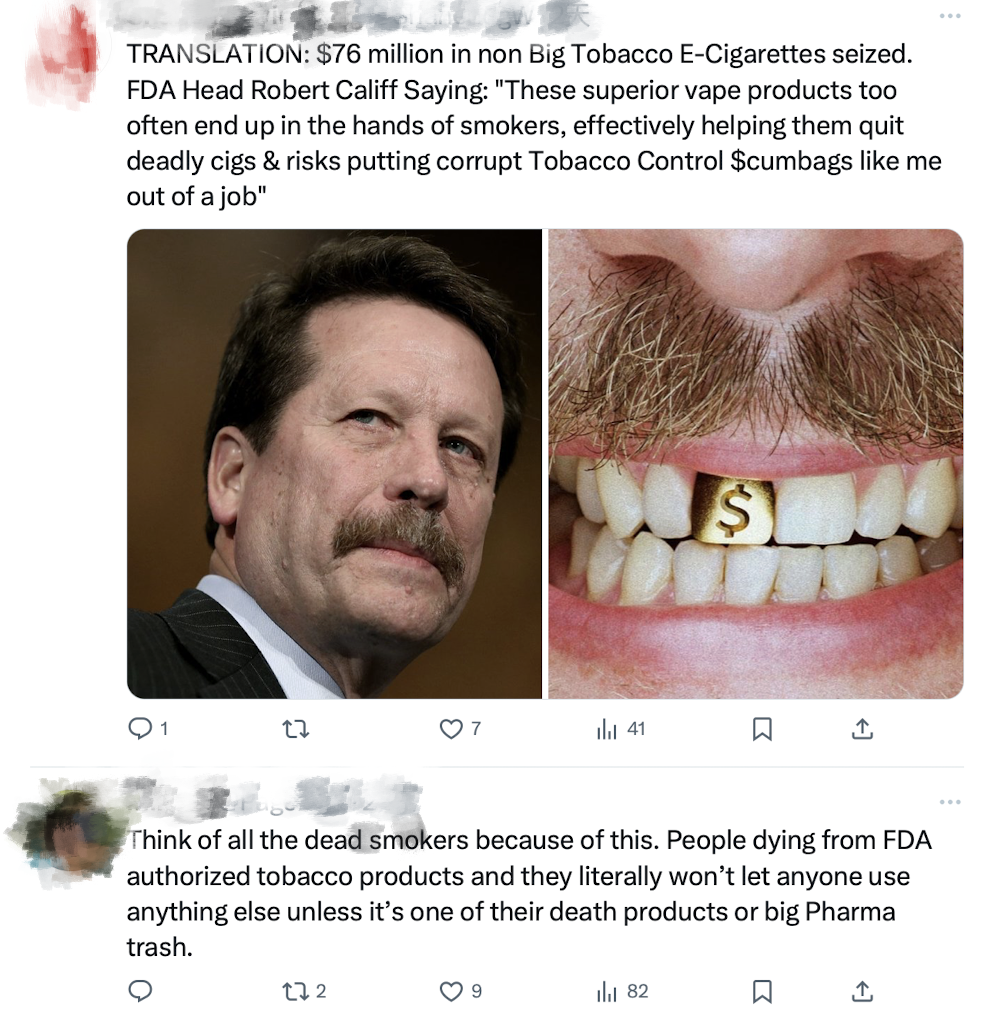
Some argue that the seizure of illegal e-cigarette products actually serves to protect the interests of big tobacco by reducing competition in the market.
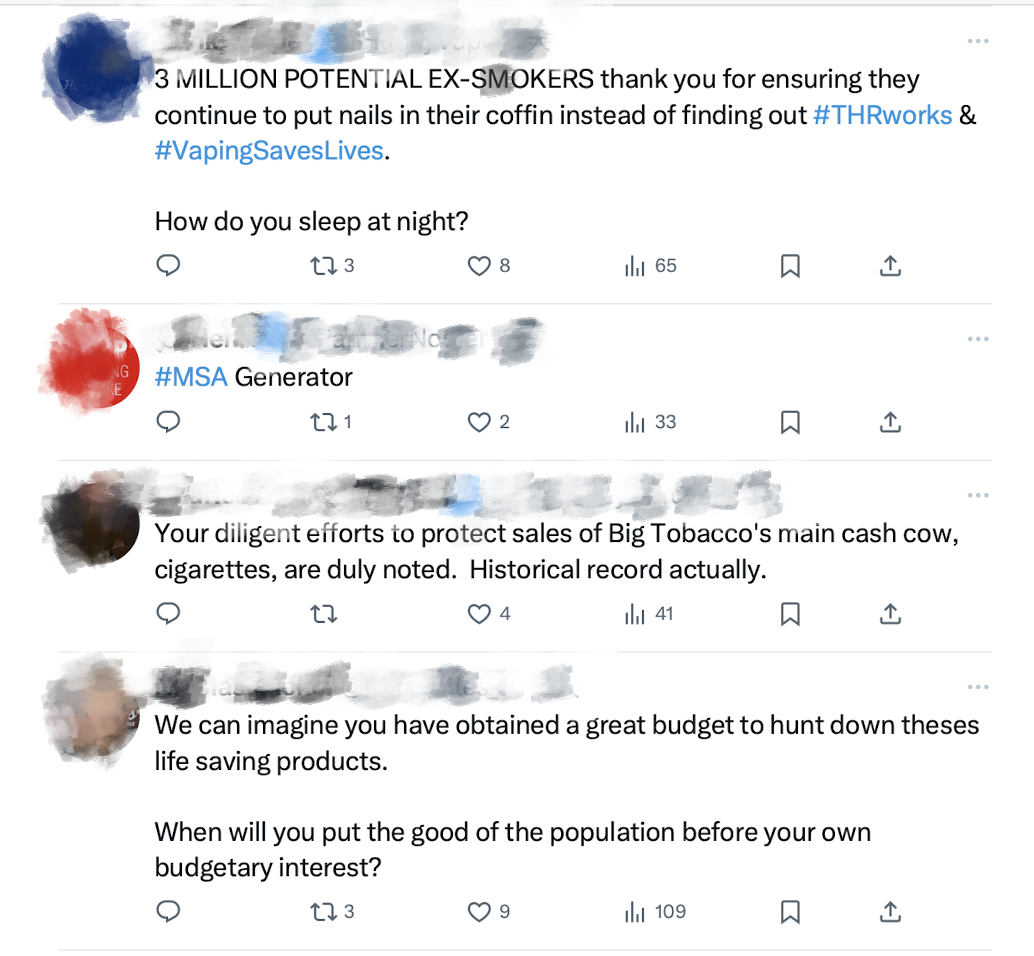
"Three million potential ex-smokers thank you for making sure they keep hammering nails into their coffins instead of discovering that #harmreduction products work and #vaping saves lives."
"Your efforts to protect Big Tobacco's main cash cow - cigarette sales - have been duly noted. This is one for the history books."
"We can only imagine the budget increase you've received to track down these life-saving products. When will you put the interests of the people before your own budgetary gains?"

We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com









