
Special Statement:
This article is limited to research on global industrial development and does not make any recommendations regarding brands, products, or investments.
Minors are prohibited from visiting.
On April 25th, 2FIRSTS discovered on the official UK website of ELFBAR that they are soon releasing a new product called ELFBAR 1200. While the product is currently listed as "coming soon" and not all information is available, some online e-cigarette stores in the UK have started revealing details. The e-cigarette website vapourcore has announced that the kit price for ELFBAR 1200 will be £6.95.
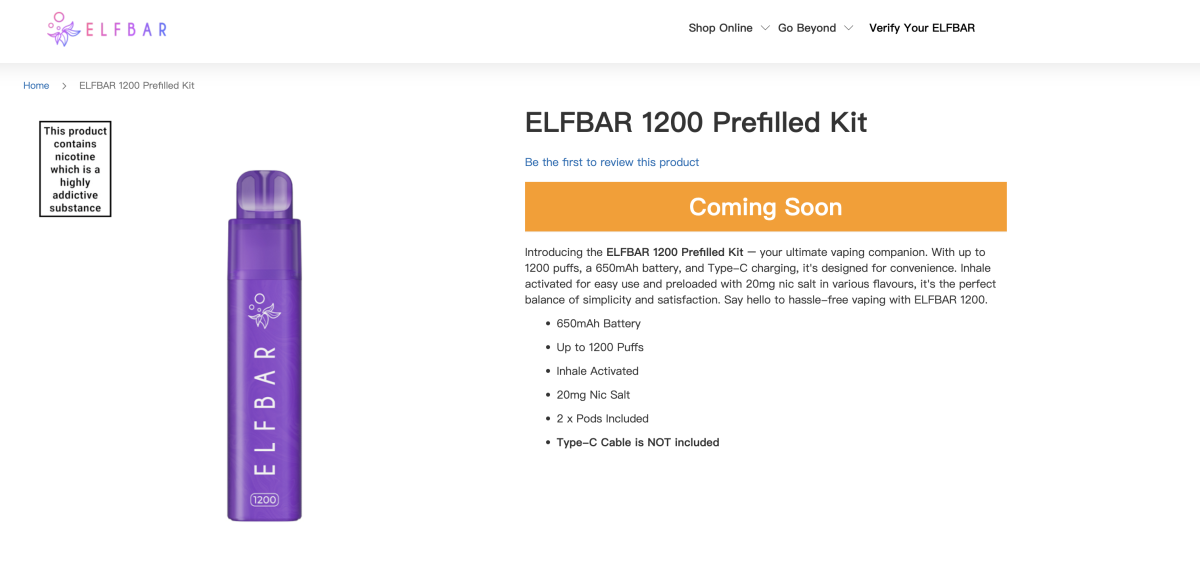
The popular e-cigarette website VAPEUK introduces the ELFBAR 1200 as a rechargeable pre-filled mini vape kit. In compliance with TPD regulations, it combines a "2-IN-1" feature, claiming to have created a legal 1200-puff disposable device in the UK. It is equipped with a 650 mAh battery, Type-C port, and QUAQ mesh coils.

It is worth noting that the device comes with two pods containing 2% nicotine and 2ml each, totaling up to 1200 puffs. This also means that users can choose two different pods for a flavor mix. It is reported that the ELFBAR 1200 offers 22 flavor options.
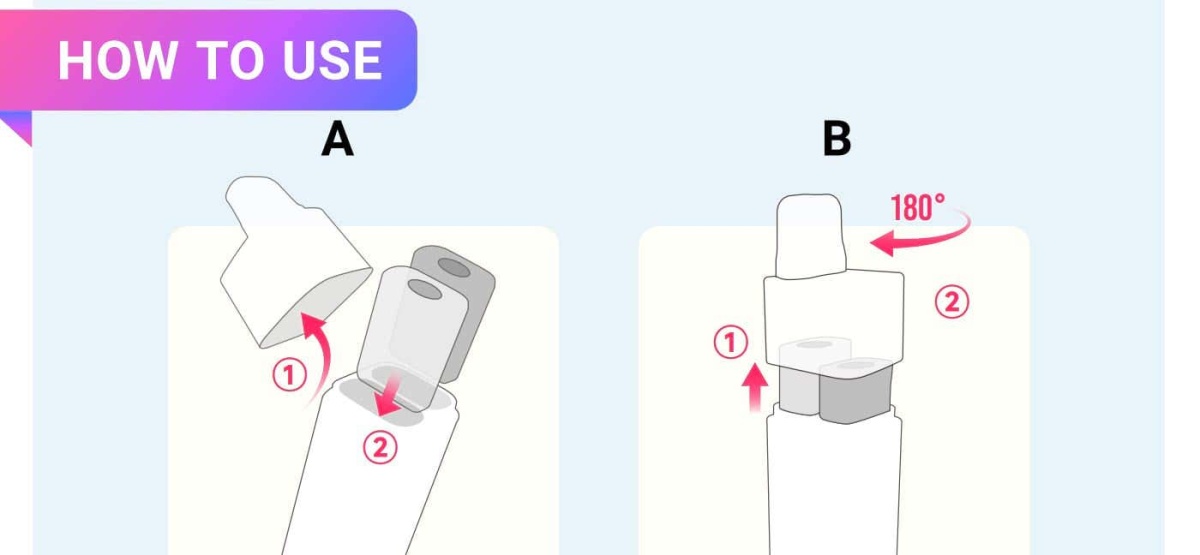
The main feature of the product is its unique installation method. Unlike a standalone pod system, users must insert the pod into two ports on the device and then attach the mouthpiece, allowing time for the e-liquid to be absorbed before use.
Products such as the IVG 2400 and Sky Hunter 2600, which feature a combination of pods to increase capacity, are not uncommon in the UK market. By using multiple pods, these products not only comply with UK regulations but also have a capacity of over 600 puffs.

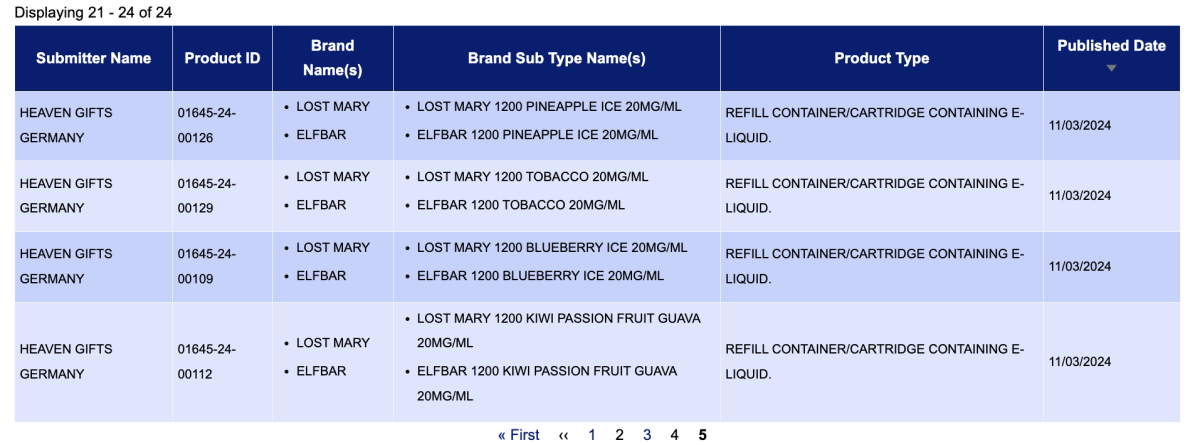
The product "ELFBAR 1200" "EB 1200" was searched using the keyword in the e-cigarette search system of the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK. It was found that the product was publicly listed on March 11, and is categorized as a "refillable container/pod filled with electronic liquid." According to UK e-cigarette regulations, this e-cigarette product has been certified and approved by the British government.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










