
Special announcement:
This article is for internal industry communication only and does not make any recommendations for specific brands or products.
The images presented in this article are only used to describe the facts and are not intended as advertisements for any products.
Minors are prohibited from accessing this article.
Key points of focus:
Market Update: Between August 18th and 24th, the UK Medicines and Healthcare products Regulatory Agency (MHRA) database listed a total of 193 e-cigarette SKUs, featuring brands such as VAPORESSO, IJOY, ALD, ASPIRE, HAYATI, and SMOK.
Mainstream product categories: The SKUs disclosed this week mainly focus on pre-filled oil pods (156 items), pre-filled oil e-cigarettes (12 items), and refillable/open system (10 items) within these three major categories.
Business news update: Several new devices from the VAPORESSO brand, a subsidiary of Smoore, have been announced, including the VAPORESSO XROS 5 MINI KIT and VAPORESSO XROS 5 Kit. Additionally, companies such as ALD and UWELL have also unveiled their latest devices and pod products.
Market Trend Analysis: The number of pre-registered oil pods announced this week is 156, a significant decrease compared to the previous week (August 11th to 17th), indicating a possible short-term fluctuation in compliance announcements for this category of products.
According to the compliance process in the UK, e-cigarette products must be listed in the Medicines and Healthcare products Regulatory Agency (MHRA) notification database. Once listed, it signifies that these SKUs have passed compliance checks and have obtained permission to be legally sold on the UK market.
2Firsts regularly compiles and analyzes relevant information from MHRA notifications to help industry professionals understand the approval status of new products in the UK market.
The following is an update of announcements made between August 18th and 24th, covering major brands, product types, and preliminary market trend analysis.
During this time period, the MHRA has publicized a total of 193 SKUs, involving brands such as VAPORESSO, IJOY, ALD, ASPIRE, HAYATI, SMOK, and others.
A total of 156 SKUs were updated from August 18 to 24, a decrease of 86.5% compared to the previous week (August 11 to 17) when 1156 SKUs were updated.
QUIXOTIC and HAYATI have updated their e-cigarette devices, with a reduced number of rechargeable products in their lineup.
In the product category of "Electronic cigarette - Rechargeable, placed on the market with one type of e-liquid (fixed combination). Any rechargeable which can also be used as a refillable should be reported under the refillable category," a total of 12 SKUs were updated between August 18th and 24th, all from the brand QUIXOTIC.
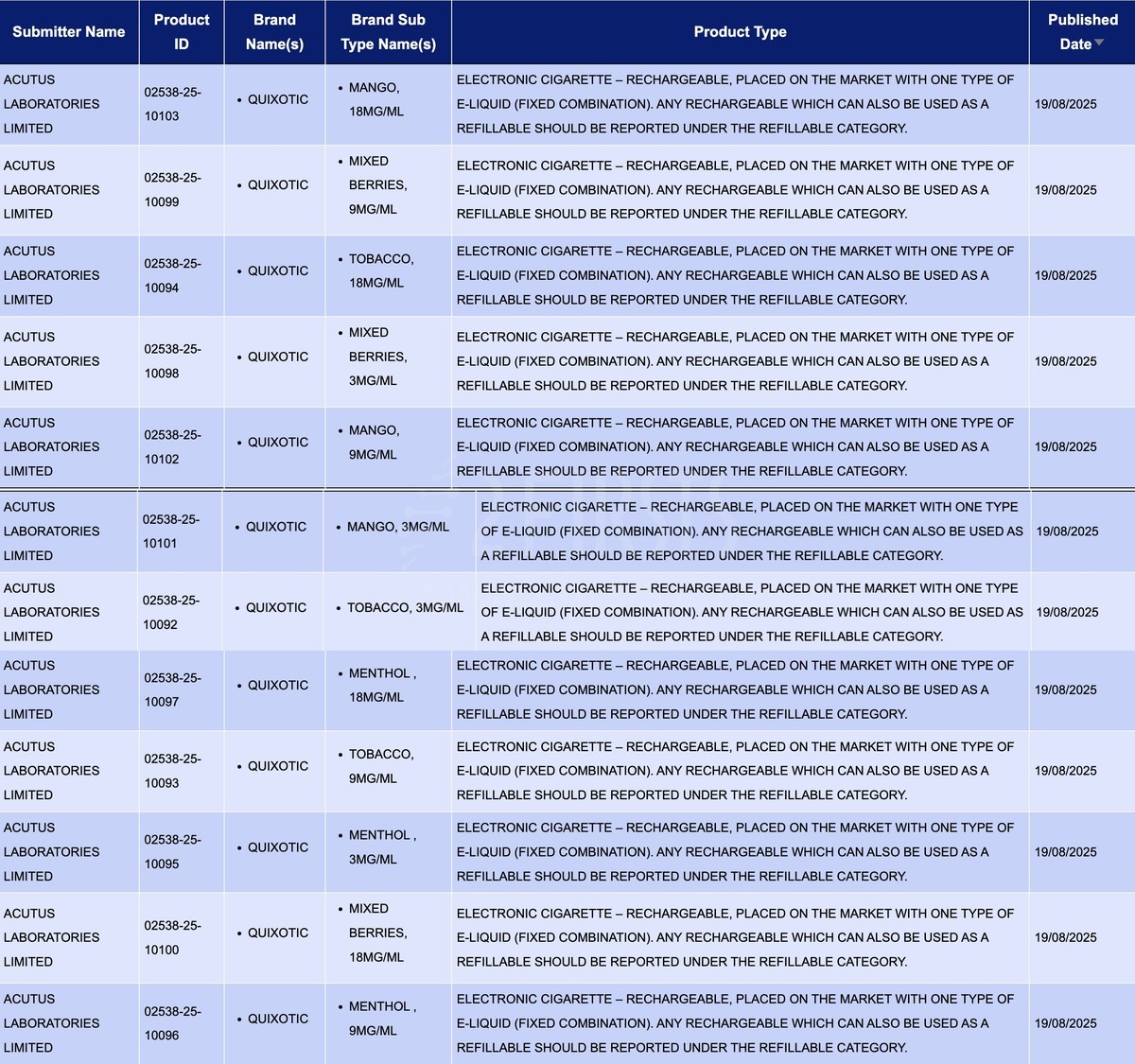
The applicant is ACUTUS LABORATORIES LIMITED, offering products in four flavors: MANGO, TOBACCO, MENTHOL, MIXD BERRIES. These flavors are available in nicotine concentrations of 3mg/ml, 9mg/ml, and 12mg/ml.
In the "Electronic cigarette - Rechargeable, device only" product category, there was a total of 1 SKU update from the HAYATI brand between August 18th and 24th.
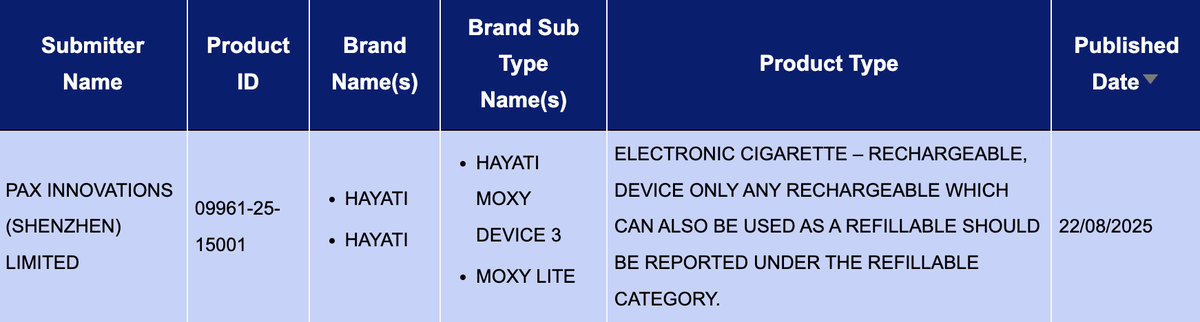
Parkes Innovation (Shenzhen) Technology Co., Ltd.'s brand HAYATI has released two devices, HAYATI Moxy Device 3 and Moxy Lite, which will be showcased as the same SKU.
Prior to this, the brand's Moxy device 1 and Moxy Pro had also been officially announced on August 12th.
IJOY, ALD, and VAPORESSO have all announced updates to their open pod systems.
In the product category of "Electronic cigarette – Refillable, placed on the market with one type of e-liquid (fixed combination)," a total of 10 SKUs were updated between August 18 and 24, all of which were from the IJOY brand.
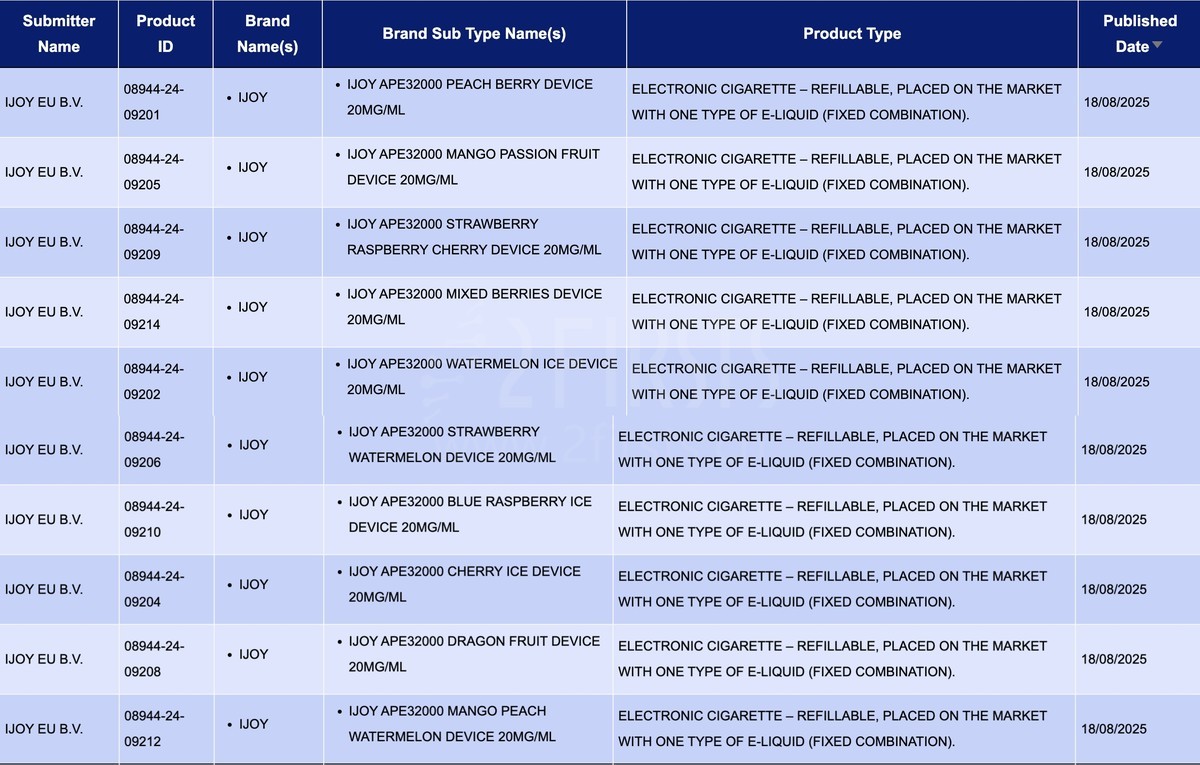
The applicant is IJOY EU B.V., the 10 flavors of IJOY APE32000 have been publicly listed, all with a nicotine concentration of 20mg/ml. It is reported that the IJOY brand is affiliated with Shenzhen Aze Technology Co., Ltd.
In the product category of "Electronic cigarette - Refillable, device only," there were a total of 3 SKU updates from August 18th to 24th, involving the brands ALD, VAPORESSO, and BIG BAR.
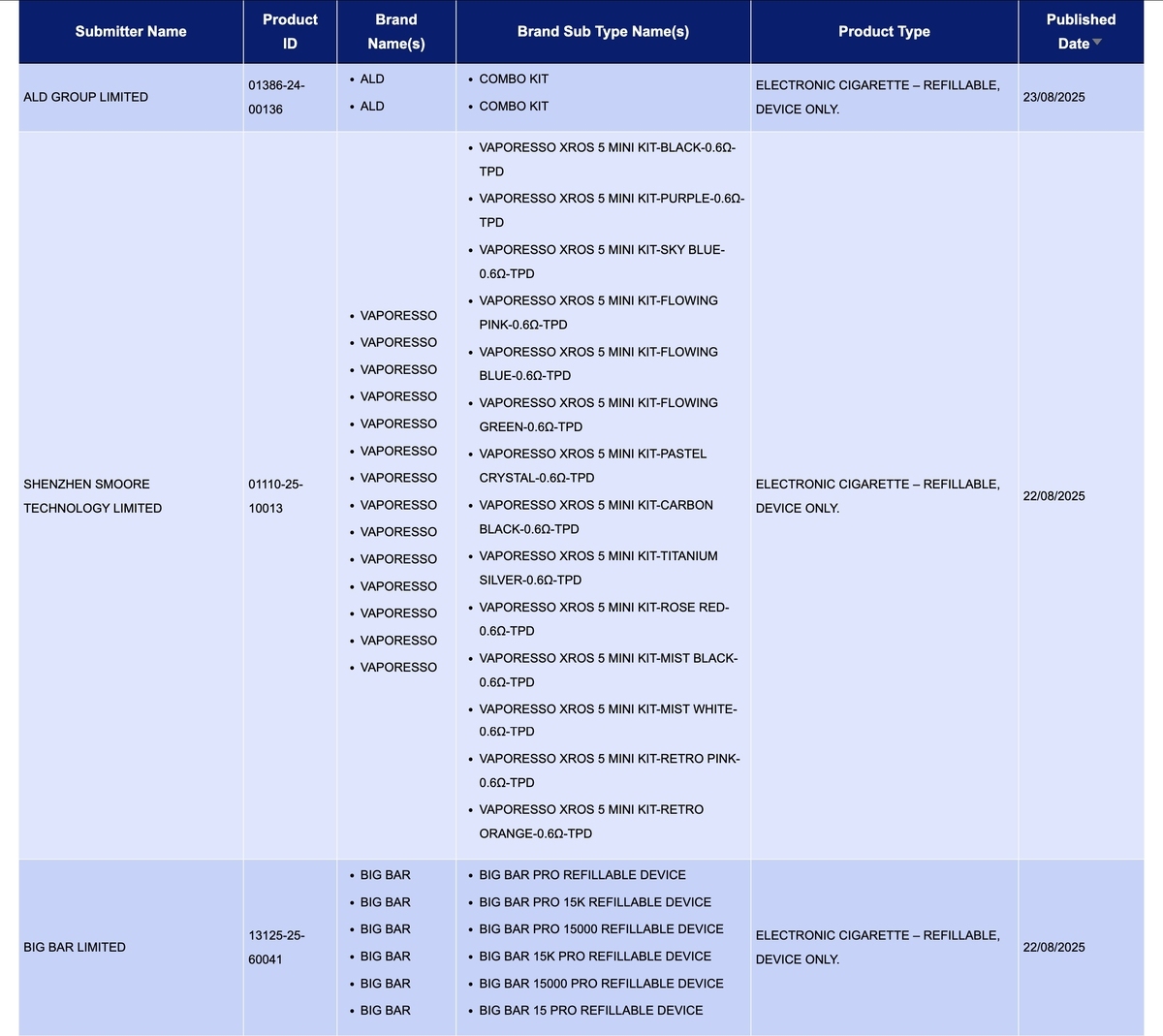
Shenzhen-based ALD Technology Co., Ltd. has unveiled its latest product, the Combo Kit under its brand ALD.
Smoore International Holdings Limited's brand VAPORESSO has released the VAPORESSO XROS 5 MINI KIT. The release includes one SKU with 14 different colored devices, all with a resistance value of 0.6Ω.
The VAPORESSO XROS 5 MINI KIT has been launched on the official VAPORESSO brand website. This product is compatible with the XROS pod and features a 1500mAh battery capacity.
As the applicant, BIG BAR LIMITED is applying for public disclosure for the brand BIG BAR PRO.
Vaporesso products are now available on the official website, while Smok is launching new products.
In the product category of "Kit – Pack containing more than one different e-cigarette device and/or more than one different refill container/cartridge," there were a total of 3 SKU updates from August 18th to 24th, involving the brands VAPORESSO, SXMINI, and SMOK.
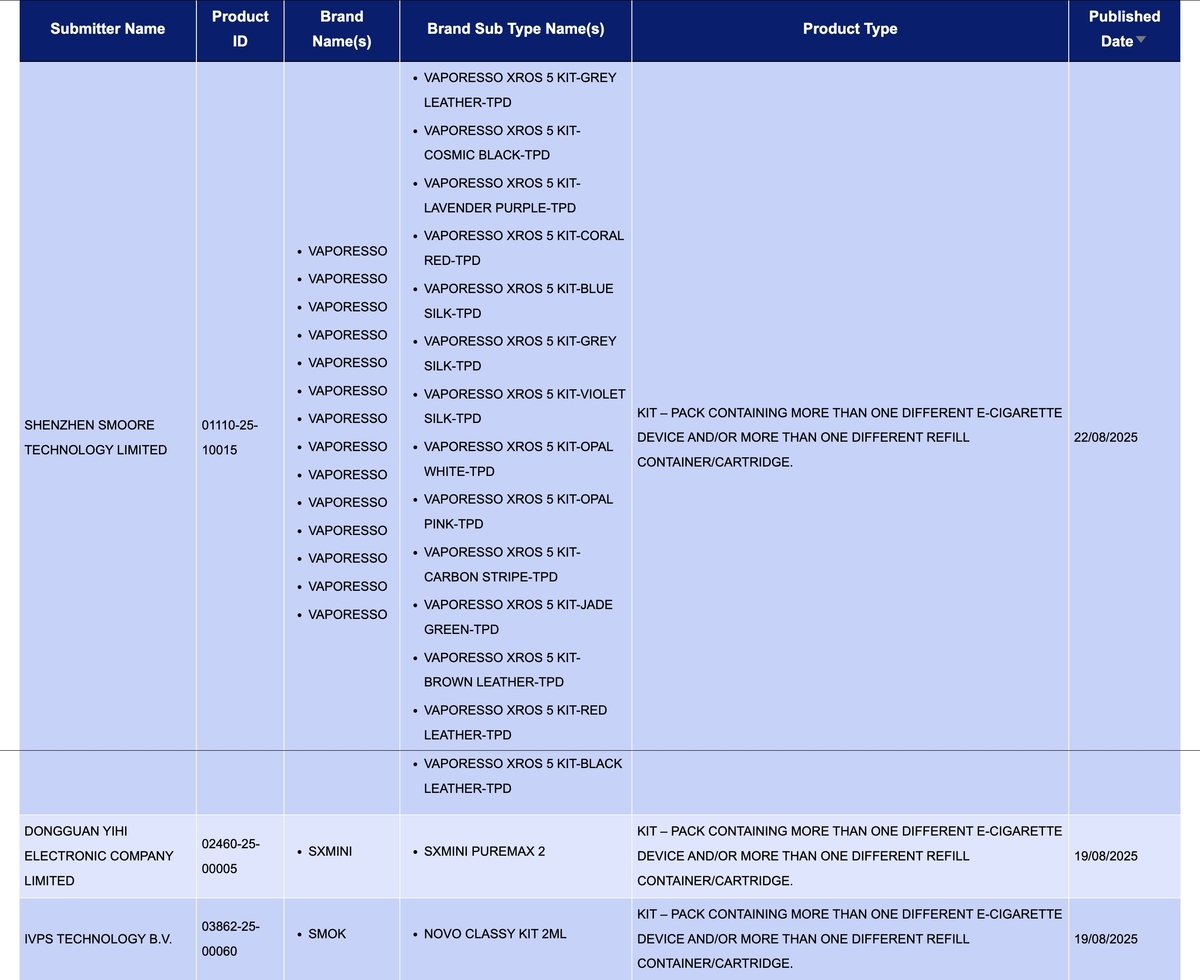
Smoore International Holdings Limited has officially launched the VAPORESSO XROS 5 Kit under its brand VAPORESSO. This release includes one SKU with 14 different colors of devices.
The VAPORESSO XROS 5 Kit has been launched on the official website of the VAPORESSO brand. This product features a 1500mAh battery and a 0.88-inch screen.
Dongguan Yihai Electronics Co., Ltd., as the applicant for the SXmini Puremax 2 under the SXMINI brand, has had its application published. The SXmini Puremax 2 features a 1300mAh battery and a 0.96-inch TFT screen, with a retail price of approximately $34.99.
The NOVO CLASSY KIT 2ml, a new product from SMOK, a brand under Shenzhen IVPS Technology Co., Ltd., has been published.
UWELL's Multiple Pods Updated, Fewer Submissions Than Last Period
In the product category "Individual part of electronic cigarette capable of containing e-liquid," a total of 8 SKUs were updated between August 18 and 24. This included brands such as ALD, UWELL, and ASPIRE.
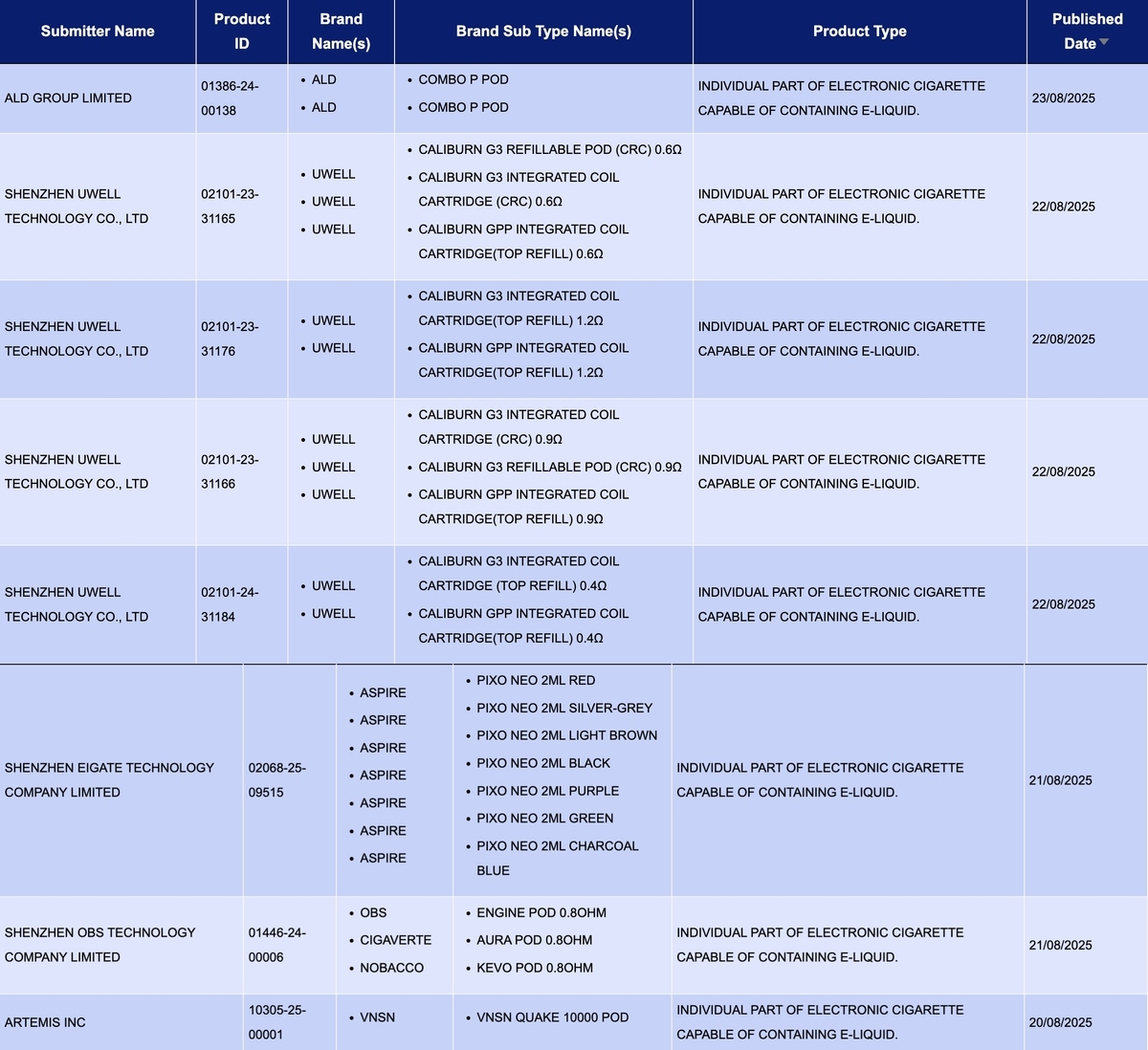
Shenzhen Zhuoli Energy Technology Co., Ltd.'s brand ALD had its Combo P Pod published.
Shenzhen Uwell Technology Co., Ltd.'s brand UWELL published multiple pod SKUs for its CALIBURN G3 and CALIBURN GPP series, covering four resistance levels: 0.4Ω, 0.6Ω, 0.9Ω, and 1.2Ω. The published products include:
- CALIBURN G3 Refillable Pod (CRC) 0.6Ω / CALIBURN G3 Integrated Coil Cartridge (CRC) 0.6Ω / CALIBURN GPP Integrated Coil Cartridge (top refill) 0.6Ω
- CALIBURN G3 Integrated Coil Cartridge (top refill) 1.2Ω / CALIBURN GPP Integrated Coil Cartridge (top refill) 1.2Ω
- CALIBURN G3 Integrated Coil Cartridge (CRC) 0.9Ω / CALIBURN G3 Refillable Pod (CRC) 0.9Ω / CALIBURN GPP Integrated Coil Cartridge (top refill) 0.9Ω
- CALIBURN G3 Integrated Coil Cartridge (top refill) 0.4Ω / CALIBURN GPP Integrated Coil Cartridge (top refill) 0.4Ω
Pixo Neo 2ml from ASPIRE, a brand under Shenzhen Eigate Technology Co., Ltd., has been published. It is available in 7 different colors.
Shenzhen Obots Technology Co., Ltd., as the applicant, submitted applications for the following products:
- OBS: Engine Pod 0.8ohm
- Cigaverte: Aura Pod 0.8ohm
- NOBACCO: KEVO POD 0.8ohm
ARTEMIS INC., as the applicant, submitted an application for the VNSN QUAKE 10000 pod from the YNSN brand.
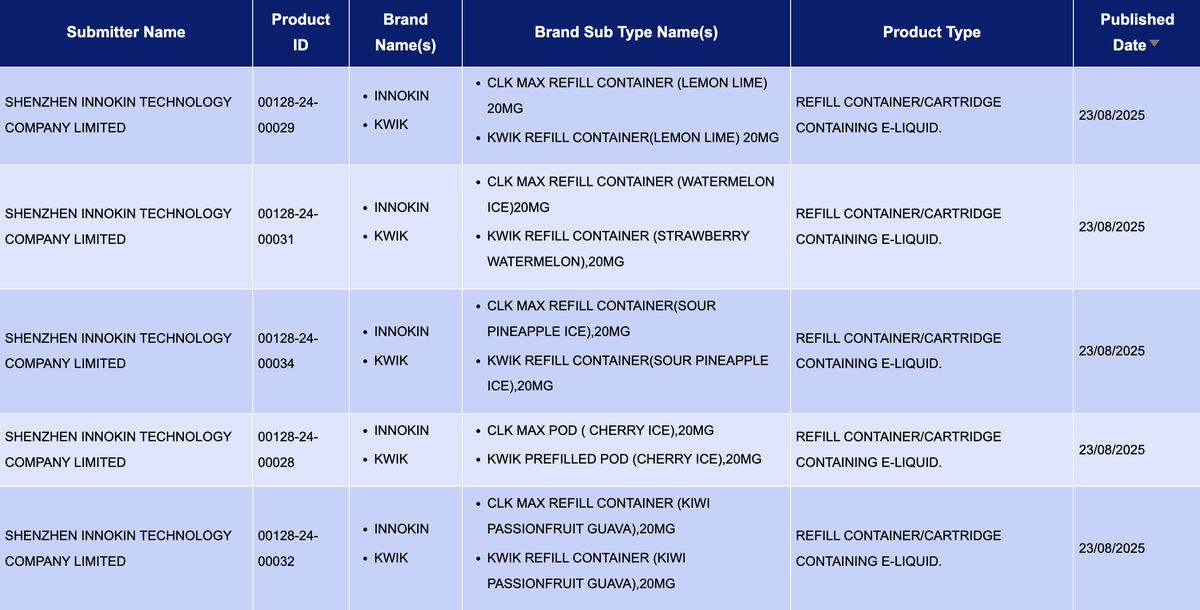
In the product category "Refill container/cartridge containing e-liquid," a total of 156 SKUs were updated between August 18 and 24, including brands like ALD, UWELL, and ASPIRE. Compared to the 1,156 pods published last week (August 11-17), the number of pod publications this week has significantly dropped.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com









